A Revised Estimate of Costs Associated With Routine Preoperative Testing in Medicare Cataract Patients With a Procedure-Specific Indicator
- PMID: 29346472
- PMCID: PMC5885904
- DOI: 10.1001/jamaophthalmol.2017.6372
A Revised Estimate of Costs Associated With Routine Preoperative Testing in Medicare Cataract Patients With a Procedure-Specific Indicator
Abstract
Importance: Routine preoperative medical testing is not recommended for patients undergoing low-risk surgery, but testing is common before surgery. A 30-day preoperative testing window is conventionally used for study purposes; however, the extent of routine testing that occurs prior to that point is unknown.
Objective: To improve on existing cost estimates by identifying all routine preoperative testing that takes place after the decision is made to perform cataract surgery.
Design, setting, and participants: This cross-sectional study assessed preoperative care in a 50% sample of Medicare beneficiaries older than 66 years who underwent ambulatory cataract surgery in 2011. Data analysis was completed from March 2016 to October 2017.
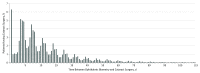
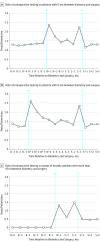
Main outcomes and measures: Using ocular biometry as a procedure-specific indicator to mark the start of the routine preoperative testing window, we measured testing rates in the interval between ocular biometry and cataract surgery and compared this with testing rates in the 6 months preceding biometry. We estimated the total cost of testing that occurred between biometry and cataract surgery.
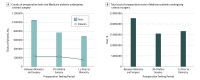
Results: A total of 440 857 patients underwent cataract surgery. A total of 423 710 (96.1%) had an ocular biometry claim before index surgery, of whom 264 514 (60.0%) were female; the mean (SD) age of the cohort was 76.1 (6.2) years. A total of 111 998 (25.4%) underwent surgery more than 30 days after biometry. Among patients with a biometry claim, the mean number of tests/patient/month increased from 1.1 in the baseline period to 1.7 in the interval between biometry and cataract surgery. Although preoperative testing peaked in all patients in the 30 days preceding surgery (1.8 tests/patient/month), the subset of patients with no overlap between postbiometry and presurgery periods experienced increased testing rates to 1.8 tests per patient per month in the 30 days after biometry, regardless of the elapsed time between biometry and surgery. The total estimated cost of routine preoperative testing in the full cohort was $22.7 million; we estimate that routine preoperative testing costs Medicare up to $45.4 million annually.
Conclusions and relevance: In this study of Medicare beneficiaries, routine preoperative medical testing occurs more often and is costlier than has been reported previously. Extra costs are attributable to testing that occurs prior to the 30-day window preceding surgery. As a cost-cutting measure, routine preoperative medical testing should be avoided in patients with cataracts throughout the interval between ocular biometry and cataract surgery.
Conflict of interest statement
Figures

Comment in
-
Preoperative Evaluations for Cataract Surgery Are Routine but Anachronistic.JAMA Ophthalmol. 2018 Mar 1;136(3):239. doi: 10.1001/jamaophthalmol.2017.6278. JAMA Ophthalmol. 2018. PMID: 29346487 No abstract available.
References
-
- The Royal College of Ophthalmologists Cataract surgery guidelines. https://www.rcophth.ac.uk/wp-content/uploads/2014/12/2010-SCI-069-Catara.... Accessed June 20, 2016.
-
- Apfelbaum JL, Connis RT, Nickinovich DG, et al. ; Committee on Standards and Practice Parameters; American Society of Anesthesiologists Task Force on Preanesthesia Evaluation . Practice advisory for preanesthesia evaluation: an updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Anesthesiology. 2012;116(3):522-538. - PubMed
-
- American Academy of Ophthalmology Routine preoperative laboratory testing for patients scheduled for cataract surgery—2014. https://www.aao.org/clinical-statement/routine-preoperative-laboratory-t.... Accessed May 20, 2015.
-
- American Academy of Ophthalmology Cataract and Anterior Segment Panel Preferred practice pattern guidelines: cataract in the adult eye. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp... Published 2016. Accessed December 11, 2017.
-
- Onuoha OC, Arkoosh VA, Fleisher LA. Choosing wisely in anesthesiology: the gap between evidence and practice. JAMA Intern Med. 2014;174(8):1391-1395. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

