Proliferation of hepatic stellate cells, mediated by YAP and TAZ, contributes to liver repair and regeneration after liver ischemia-reperfusion injury
- PMID: 29351389
- PMCID: PMC5966748
- DOI: 10.1152/ajpgi.00153.2017
Proliferation of hepatic stellate cells, mediated by YAP and TAZ, contributes to liver repair and regeneration after liver ischemia-reperfusion injury
Abstract
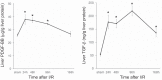
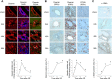
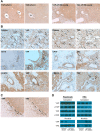
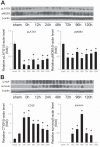
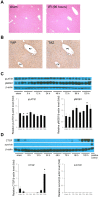
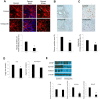
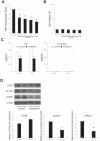
Yes-associated protein (YAP) and transcriptional coactivator with PDZ-binding motif (TAZ) are key regulators of cell proliferation and organ size; however, their physiological contribution after liver injury has not been fully understood. In this study, we sought to determine the role of YAP and TAZ during liver recovery after ischemia-reperfusion (I/R). A murine model of partial (70%) I/R was used to induce liver injury and study the reparative and regenerative response. After liver injury, there was marked activation and proliferation of hepatic stellate cells. The Hippo pathway components, large tumor suppressor 1 (LATS1) and its adapter protein, Mps one binder 1 (MOB1), were inactivated during liver repair, and YAP and TAZ were activated selectively in hepatic stellate cells. Concurrently, the expression of connective tissue growth factor and survivin, both of which are YAP and TAZ target genes, were upregulated. Hepatic stellate cell expansion and concomitant activation of YAP and TAZ occurred only in the injured liver and were not observed in the nonischemic liver. Treatment of mice with verteporfin, an inhibitor of YAP and TAZ, decreased hepatic stellate cell proliferation, survivin, and cardiac ankyrin repeat protein expression. These changes were associated with a significant decrease in hepatocyte proliferation. The data suggest that liver repair and regeneration after I/R injury are dependent on hepatic stellate cell proliferation, which is mediated by YAP and TAZ. NEW & NOTEWORTHY This study is the first to assess the proliferation of hepatic stellate cells (HSCs) after ischemia-reperfusion (I/R) injury and their role in the reparative and regenerative process. Here we show that the Hippo pathway is inactivated after I/R and that Yes-associated protein/transcriptional coactivator with PDZ-binding motif (YAP/TAZ) activation is detected in HSC. HSC proliferation and expansion are prominent during liver recovery after I/R injury. Inhibition of YAP/TAZ activation with verteporfin reduces HSC proliferation and target gene expression and attenuates hepatocyte proliferation.
Keywords: hepatic stellate cell proliferation; liver injury; liver regeneration; survivin.
Figures

References
-
- Baba HA, Wohlschlaeger J, Schmitz KJ, Nadalin S, Lang H, Benesch A, Gu Y, Biglarnia AR, Sotiropoulos GC, Takeda A, Takeda N, von Wnuck Lipinski K, Levkau B. Survivin is upregulated during liver regeneration in rats and humans and is associated with hepatocyte proliferation. Liver Int 29: 585–592, 2009. doi: 10.1111/j.1478-3231.2008.01911.x. - DOI - PubMed
-
- Barone S, Okaya T, Rudich S, Petrovic S, Tenrani K, Wang Z, Zahedi K, Casero RA, Lentsch AB, Soleimani M. Distinct and sequential upregulation of genes regulating cell growth and cell cycle progression during hepatic ischemia-reperfusion injury. Am J Physiol Cell Physiol 289: C826–C835, 2005. doi: 10.1152/ajpcell.00629.2004. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

