Core Differences in Synaptic Signaling Between Primary Visual and Dorsolateral Prefrontal Cortex
- PMID: 29351585
- PMCID: PMC6041807
- DOI: 10.1093/cercor/bhx357
Core Differences in Synaptic Signaling Between Primary Visual and Dorsolateral Prefrontal Cortex
Abstract
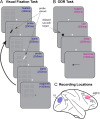
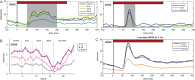
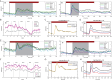
Neurons in primary visual cortex (V1) are more resilient than those in dorsolateral prefrontal cortex (dlPFC) in aging, schizophrenia and Alzheimer's disease. The current study compared glutamate and neuromodulatory actions in macaque V1 to those in dlPFC, and found striking regional differences. V1 neuronal firing to visual stimuli depended on AMPA receptors, with subtle NMDA receptor contributions, while dlPFC depends primarily on NMDA receptors. Neuromodulatory actions also differed between regions. In V1, cAMP signaling increased neuronal firing, and the phosphodiesterase PDE4A was positioned to regulate cAMP effects on glutamate release from axons. HCN channels in V1 were classically located on distal dendrites, and enhanced cell firing. These data contrast with dlPFC, where PDE4A and HCN channels are concentrated in thin spines, and cAMP-HCN signaling gates inputs and weakens firing. These regional differences may explain why V1 neurons are more resilient than dlPFC neurons to the challenges of age and disease.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

