Estrogen Receptor-Positive Breast Cancer: Exploiting Signaling Pathways Implicated in Endocrine Resistance
- PMID: 29352052
- PMCID: PMC5947450
- DOI: 10.1634/theoncologist.2017-0423
Estrogen Receptor-Positive Breast Cancer: Exploiting Signaling Pathways Implicated in Endocrine Resistance
Abstract
Advancements in molecular profiling and endocrine therapy (ET) have led to more focused clinical attention on precision medicine. These advances have expanded our understanding of breast cancer (BC) pathogenesis and hold promising implications for the future of therapy. The estrogen receptor-α is a predominant endocrine regulatory protein in the breast and in estrogen-induced BC. Successful targeting of proteins and genes within estrogen receptor (ER) nuclear and nonnuclear pathways remains a clinical goal. Several classes of antiestrogenic agents are available for patients with early, advanced, or metastatic BC, including selective ER modulators, aromatase inhibitors, and a selective ER degrader. Clinical development is focused upon characterizing the efficacy and tolerability of inhibitors that target the phosphatidylinositol 3 kinase (PI3K)/akt murine thymoma viral oncogene (AKT)/mammalian target of rapamycin inhibitor (mTOR) signaling pathway or the cyclin-dependent kinase 4/6 (CDK4/6) cell cycle pathway in women with hormone receptor-positive, human epidermal growth receptor 2-negative BC who have demonstrated disease recurrence or progression. De novo and acquired resistance remain a major challenge for women with BC receiving antiestrogenic therapy. Therefore, sequential combination of targeted ET is preferred in these patients, and the ever-increasing understanding of resistance mechanisms may better inform the selection of future therapy. This review describes the intricate roles of the PI3K/AKT/mTOR and CDK4/6 pathways in intracellular signaling and the use of endocrine and endocrine-based combination therapy in BC.
Implications for practice: The foundational strategy for treating hormone receptor-positive, human epidermal growth receptor 2-negative, advanced breast cancer includes the use of endocrine therapy either alone or in combination with targeted agents. The use of combination therapy aims to downregulate cell-signaling pathways with the intent of minimizing cellular "crosstalk," which can otherwise result in continued tumorigenesis or progression through redundant pathways. This review provides the clinician with the molecular rationale and clinical evidence for these treatments and refers to evidence-based guidelines to inform the decision-making process.
分子表达谱和内分泌疗法 (ET) 的进步使得精准医疗获得更为集中的临床关注。这些进步拓展了我们对乳腺癌 (BC) 发病机制的了解, 并预示未来疗法的良好前景。雌激素受体 α 是乳腺及雌激素诱导 BC 中的主要内分泌调节蛋白。对雌激素受体 (ER) 核途径和非核途径中的蛋白质和基因进行成功靶向仍是临床目标。早期、晚期和转移性 BC 患者现可使用多种抗雌激素药物, 包括选择性 ER 调节剂、芳香化酶抑制剂和选择性 ER 降解剂。临床开发专注于表征以磷脂酰肌醇 3 激酶 (PI3K)/akt 鼠胸腺瘤病毒致癌基因 (AKT)/哺乳动物雷帕霉素靶蛋白 (mTOR) 抑制剂信号传导通路或细胞周期依赖性激酶 4/6 (CDK4/6) 细胞周期通路为靶标的抑制剂在显现疾病复发或进展的激素受体阳性、人表皮生长受体 2 阴性 BC 女性中的疗效和耐受性。在接受抗雌激素治疗的 BC 女性中, 原发性及获得性耐药仍是主要挑战。因此, 靶向 ET 的序贯联合治疗是这些患者的首选, 对耐药性机制的理解不断增加可更好地指导未来疗法的选择。本综述介绍了 PI3K/AKT/mTOR 和 CDK4/6 通路在细胞内信号传导中的复杂作用以及内分泌和以内分泌为基础的联合疗法在 BC 中的使用。
对临床实践的启示:激素受体阳性、人表皮生长受体 2 阴性晚期乳腺癌的基本治疗策略包括单独使用内分泌疗法或与靶向药物联用。使用联合疗法旨在下调细胞信号传导通路, 目的最大程度地减少细胞“串扰”, 否则可能会通过冗余途径导致持续肿瘤发生或进展。本综述为临床医生提供了该类治疗的分子依据和临床证据, 并援引循证指南指导决策过程
Keywords: Aromatase inhibitor; CDK4/6 inhibitor; Endocrine therapy; Estrogen receptor degrader; mTOR inhibitor.
© AlphaMed Press 2018.
Conflict of interest statement
Disclosures of potential conflicts of interest may be found at the end of this article.
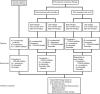
Figures

References
-
- Murphy CG, Dickler MN. Endocrine resistance in hormone‐responsive breast cancer: Mechanisms and therapeutic strategies. Endocr Relat Cancer 2016;23:R337–R352. - PubMed
-
- National Comprehensive Cancer Network . NCCN Clinical Practice Guidelines in Oncology: Breast Cancer. Version 3.2017. November 10, 2017. Available at https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf. Accessed November 16, 2017.
-
- Robertson JF, Bondarenko IM, Trishkina E et al. Fulvestrant 500 mg versus anastrozole 1 mg for hormone receptor‐positive advanced breast cancer (FALCON): An international, randomised, double‐blind, phase 3 trial. Lancet 2017;388:2997–3005. - PubMed
-
- Rugo HS, Rumble RB, Macrae E et al. Endocrine therapy for hormone receptor‐positive metastatic breast cancer: American Society of Clinical Oncology guideline . J Clin Oncol 2016;34:3069–3103. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

