Expression and relevance of the G protein-gated K+ channel in the mouse ventricle
- PMID: 29352184
- PMCID: PMC5775354
- DOI: 10.1038/s41598-018-19719-x
Expression and relevance of the G protein-gated K+ channel in the mouse ventricle
Abstract
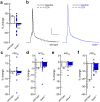
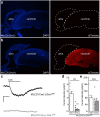
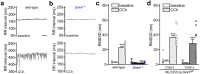
The atrial G protein-gated inwardly rectifying K+ (GIRK) channel is a critical mediator of parasympathetic influence on cardiac physiology. Here, we probed the details and relevance of the GIRK channel in mouse ventricle. mRNAs for the atrial GIRK channel subunits (GIRK1, GIRK4), M2 muscarinic receptor (M2R), and RGS6, a negative regulator of atrial GIRK-dependent signaling, were detected in mouse ventricle at relatively low levels. The cholinergic agonist carbachol (CCh) activated small GIRK currents in adult wild-type ventricular myocytes that exhibited relatively slow kinetics and low CCh sensitivity; these currents were absent in ventricular myocytes from Girk1-/- or Girk4-/- mice. While loss of GIRK channels attenuated the CCh-induced shortening of action potential duration and suppression of ventricular myocyte excitability, selective ablation of GIRK channels in ventricle had no effect on heart rate, heart rate variability, or electrocardiogram parameters at baseline or after CCh injection. Additionally, loss of ventricular GIRK channels did not impact susceptibility to ventricular arrhythmias. These data suggest that the mouse ventricular GIRK channel is a GIRK1/GIRK4 heteromer, and show that while it contributes to the cholinergic suppression of ventricular myocyte excitability, this influence does not substantially impact cardiac physiology or ventricular arrhythmogenesis in the mouse.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





References
-
- Schwartz PJ. The autonomic nervous system and sudden death. Eur. Heart J. 1998;19(Suppl F):F72–80. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

