Parthenolide prevents resistance of MDA-MB231 cells to doxorubicin and mitoxantrone: the role of Nrf2
- PMID: 29354292
- PMCID: PMC5712634
- DOI: 10.1038/cddiscovery.2017.78
Parthenolide prevents resistance of MDA-MB231 cells to doxorubicin and mitoxantrone: the role of Nrf2
Abstract
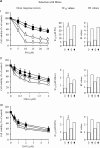
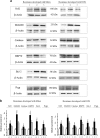
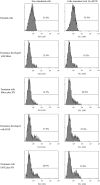
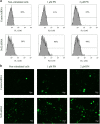
Triple-negative breast cancer is a group of aggressive cancers with poor prognosis owing to chemoresistance, recurrence and metastasis. New strategies are required that could reduce chemoresistance and increases the effectiveness of chemotherapy. The results presented in this paper, showing that parthenolide (PN) prevents drug resistance in MDA-MB231 cells, represent a contribution to one of these possible strategies. MDA-MB231 cells, the most studied line of TNBC cells, were submitted to selection treatment with mitoxantrone (Mitox) and doxorubicin (DOX). The presence of resistant cells was confirmed through the measurement of the resistance index. Cells submitted to this treatment exhibited a remarkable increment of NF-E2-related factor 2 (Nrf2) level, which was accompanied by upregulation of catalase, MnSOD, HSP70, Bcl-2 and P-glycoprotein. Moreover, as a consequence of overexpression of Nrf2 and correlated proteins, drug-treated cells exhibited a much lower ability than parental cells to generate ROS in response to a suitable stimulation. The addition of PN (2.0 μM) to Mitox and DOX, over the total selection time, prevented both the induction of resistance and the overexpression of Nrf2 and correlated proteins, whereas the cells showed a good ability to generate ROS in response to adequate stimulation. To demonstrate that Nrf2 exerted a crucial role in the induction of resistance, the cells were transiently transfected with a specific small interfering RNA for Nrf2. Similarly to the effects induced by PN, downregulation of Nrf2 was accompanied by reductions in the levels of catalase, MnSOD, HSP70 and Bcl-2, prevention of chemoresistance and increased ability to generate ROS under stimulation. In conclusion, our results show that PN inhibited the development of the resistance toward Mitox and DOX, and suggest that these effects were correlated with the prevention of the overexpression of Nrf2 and its target proteins, which occurred in the cells submitted to drug treatment.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin 2016; 66: 7–30. - PubMed
-
- Morris GJ, Naidu S, Topham AK, Guiles F, Xu Y, McCue P et al. Differences in breast carcinoma characteristics in newly diagnosed African-American and Caucasian patients: a single-institution compilation compared with the National Cancer Institute's Surveillance, Epidemiology, and End Results database. Cancer 2007; 110: 876–884. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

