JAK2/IDH-mutant-driven myeloproliferative neoplasm is sensitive to combined targeted inhibition
- PMID: 29355841
- PMCID: PMC5785272
- DOI: 10.1172/JCI94516
JAK2/IDH-mutant-driven myeloproliferative neoplasm is sensitive to combined targeted inhibition
Erratum in
-
JAK2/IDH-mutant-driven myeloproliferative neoplasm is sensitive to combined targeted inhibition.J Clin Invest. 2018 Oct 1;128(10):4743. doi: 10.1172/JCI124920. Epub 2018 Sep 17. J Clin Invest. 2018. PMID: 30222137 Free PMC article. No abstract available.
Abstract
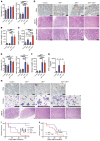
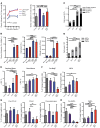
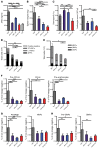
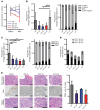
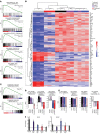
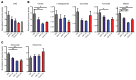
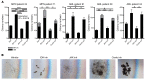
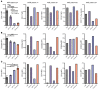
Patients with myeloproliferative neoplasms (MPNs) frequently progress to bone marrow failure or acute myeloid leukemia (AML), and mutations in epigenetic regulators such as the metabolic enzyme isocitrate dehydrogenase (IDH) are associated with poor outcomes. Here, we showed that combined expression of Jak2V617F and mutant IDH1R132H or Idh2R140Q induces MPN progression, alters stem/progenitor cell function, and impairs differentiation in mice. Jak2V617F Idh2R140Q-mutant MPNs were sensitive to small-molecule inhibition of IDH. Combined inhibition of JAK2 and IDH2 normalized the stem and progenitor cell compartments in the murine model and reduced disease burden to a greater extent than was seen with JAK inhibition alone. In addition, combined JAK2 and IDH2 inhibitor treatment also reversed aberrant gene expression in MPN stem cells and reversed the metabolite perturbations induced by concurrent JAK2 and IDH2 mutations. Combined JAK2 and IDH2 inhibitor therapy also showed cooperative efficacy in cells from MPN patients with both JAK2mut and IDH2mut mutations. Taken together, these data suggest that combined JAK and IDH inhibition may offer a therapeutic advantage in this high-risk MPN subtype.
Keywords: Drug therapy; Hematology; Leukemias; Oncology.
Conflict of interest statement
Figures
References
-
- Mesa RA, Li CY, Ketterling RP, Schroeder GS, Knudson RA, Tefferi A. Leukemic transformation in myelofibrosis with myeloid metaplasia: a single-institution experience with 91 cases. Blood. 2005;105(3):973–977. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

