Basal forebrain contributes to default mode network regulation
- PMID: 29363595
- PMCID: PMC5819396
- DOI: 10.1073/pnas.1712431115
Basal forebrain contributes to default mode network regulation
Abstract
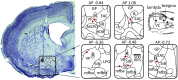
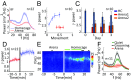
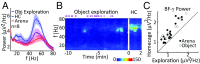
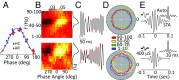
The default mode network (DMN) is a collection of cortical brain regions that is active during states of rest or quiet wakefulness in humans and other mammalian species. A pertinent characteristic of the DMN is a suppression of local field potential gamma activity during cognitive task performance as well as during engagement with external sensory stimuli. Conversely, gamma activity is elevated in the DMN during rest. Here, we document that the rat basal forebrain (BF) exhibits the same pattern of responses, namely pronounced gamma oscillations during quiet wakefulness in the home cage and suppression of this activity during active exploration of an unfamiliar environment. We show that gamma oscillations are localized to the BF and that gamma-band activity in the BF has a directional influence on a hub of the rat DMN, the anterior cingulate cortex, during DMN-dominated brain states. The BF is well known as an ascending, activating, neuromodulatory system involved in wake-sleep regulation, memory formation, and regulation of sensory information processing. Our findings suggest a hitherto undocumented role of the BF as a subcortical node of the DMN, which we speculate may be important for switching between internally and externally directed brain states. We discuss potential BF projection circuits that could underlie its role in DMN regulation and highlight that certain BF nuclei may provide potential target regions for up- or down-regulation of DMN activity that might prove useful for treatment of DMN dysfunction in conditions such as epilepsy or major depressive disorder.
Keywords: anterior cingulate cortex; gamma suppression; granger causality.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Gamma band directional interactions between basal forebrain and visual cortex during wake and sleep states.J Physiol Paris. 2016 Sep;110(1-2):19-28. doi: 10.1016/j.jphysparis.2016.11.011. Epub 2016 Nov 29. J Physiol Paris. 2016. PMID: 27913167
-
Subcortical control of the default mode network: Role of the basal forebrain and implications for neuropsychiatric disorders.Brain Res Bull. 2022 Jul;185:129-139. doi: 10.1016/j.brainresbull.2022.05.005. Epub 2022 May 11. Brain Res Bull. 2022. PMID: 35562013 Free PMC article. Review.
-
Mediodorsal thalamus and ventral pallidum contribute to subcortical regulation of the default mode network.Commun Biol. 2024 Jul 23;7(1):891. doi: 10.1038/s42003-024-06531-9. Commun Biol. 2024. PMID: 39039239 Free PMC article.
-
Ventral pallidum regulates the default mode network, controlling transitions between internally and externally guided behavior.Proc Natl Acad Sci U S A. 2021 Sep 7;118(36):e2103642118. doi: 10.1073/pnas.2103642118. Proc Natl Acad Sci U S A. 2021. PMID: 34462351 Free PMC article.
-
[Advances in the study of the effect of basal forebrain on sleep-wake regulation].Yao Xue Xue Bao. 2016 Aug;51(8):1196-201. Yao Xue Xue Bao. 2016. PMID: 29897712 Review. Chinese.
Cited by
-
Neuroimaging of traumatic brain injury in military personnel: An overview.J Clin Neurosci. 2019 Dec;70:1-10. doi: 10.1016/j.jocn.2019.07.001. Epub 2019 Jul 19. J Clin Neurosci. 2019. PMID: 31331746 Free PMC article. Review.
-
Overproduction of hydrogen sulfide, generated by cystathionine β-synthase, disrupts brain wave patterns and contributes to neurobehavioral dysfunction in a rat model of down syndrome.Redox Biol. 2022 May;51:102233. doi: 10.1016/j.redox.2022.102233. Epub 2022 Jan 13. Redox Biol. 2022. PMID: 35042677 Free PMC article.
-
Cholinergic modulation supports dynamic switching of resting state networks through selective DMN suppression.PLoS Comput Biol. 2024 Jun 6;20(6):e1012099. doi: 10.1371/journal.pcbi.1012099. eCollection 2024 Jun. PLoS Comput Biol. 2024. PMID: 38843298 Free PMC article.
-
The brain's default network: updated anatomy, physiology and evolving insights.Nat Rev Neurosci. 2019 Oct;20(10):593-608. doi: 10.1038/s41583-019-0212-7. Epub 2019 Sep 6. Nat Rev Neurosci. 2019. PMID: 31492945 Review.
-
Cholinergic modulation of spatial learning, memory and navigation.Eur J Neurosci. 2018 Sep;48(5):2199-2230. doi: 10.1111/ejn.14089. Epub 2018 Aug 19. Eur J Neurosci. 2018. PMID: 30055067 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

