Colorectal cancer (CRC) monitoring by 6-monthly 18FDG-PET/CT: an open-label multicentre randomised trial
- PMID: 29365058
- PMCID: PMC5913635
- DOI: 10.1093/annonc/mdy031
Colorectal cancer (CRC) monitoring by 6-monthly 18FDG-PET/CT: an open-label multicentre randomised trial
Abstract
Background: [18F]2-fluoro-2-deoxy-d-glucose positron emission tomography/computed tomography (18FDG-PET/CT) has high sensitivity for detecting recurrences of colorectal cancer (CRC). Our objective was to determine whether adding routine 6-monthly 18FDG-PET/CT to our usual monitoring strategy improved patient outcomes and to assess the effect on costs.
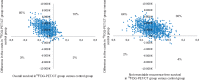
Patients and methods: In this open-label multicentre trial, patients in remission of CRC (stage II perforated, stage III, or stage IV) after curative surgery were randomly assigned (1 : 1) to usual monitoring alone (3-monthly physical and tumour marker assays, 6-monthly liver ultrasound and chest radiograph, and 6-monthly whole-body computed tomography) or with 6-monthly 18FDG-PET/CT, for 3 years. A multidisciplinary committee reviewed each patient's data every 3 months and classified the recurrence status as yes/no/doubtful. Recurrences were treated with curative surgery alone if feasible and with chemotherapy otherwise. The primary end point was treatment failure defined as unresectable recurrence or death. Relative risks were estimated, and survival was analysed using the Kaplan-Meier method, log-rank test, and Cox models. Direct costs were compared.
Results: Of the 239 enrolled patients, 120 were in the intervention arm and 119 in the control arm. The failure rate was 29.2% (31 unresectable recurrences and 4 deaths) in the intervention group and 23.7% (27 unresectable recurrences and 1 death) in the control group (relative risk = 1.23; 95% confidence interval, 0.80-1.88; P = 0.34). The multivariate analysis also showed no significant difference (hazards ratio, 1.33; 95% confidence interval, 0.8-2.19; P = 0.27). Median time to diagnosis of unresectable recurrence (months) was significantly shorter in the intervention group [7 (3-20) versus 14.3 (7.3-27), P = 0.016]. Mean cost/patient was higher in the intervention group (18 192 ± 27 679 € versus 11 131 ± 13 €, P < 0.033).
Conclusion: 18FDG-PET/CT, when added every 6 months, increased costs without decreasing treatment failure rates in patients in remission of CRC. The control group had very close follow-up, and any additional improvement (if present) would be small and hard to detect.
Clinicaltrials.gov identifier: NCT00624260.
Figures


References
-
- Rodriguez-Moranta F, Salo J, Arcusa A. et al. Postoperative surveillance in patients with colorectal cancer who have undergone curative resection: a prospective, multicenter, randomized, controlled trial. J Clin Oncol 2006; 24(3): 386–393. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

