Rosmarinic acid attenuates cardiac fibrosis following long-term pressure overload via AMPKα/Smad3 signaling
- PMID: 29367637
- PMCID: PMC5833382
- DOI: 10.1038/s41419-017-0123-3
Rosmarinic acid attenuates cardiac fibrosis following long-term pressure overload via AMPKα/Smad3 signaling
Abstract
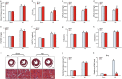
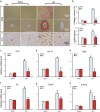
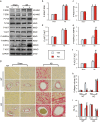
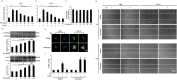
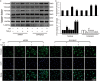
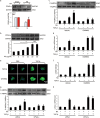
Agonists of peroxisome proliferator-activated receptor gamma (PPAR-γ) can activate 5' AMP-activated protein kinase alpha (AMPKα) and exert cardioprotective effects. A previous study has demonstrated that rosmarinic acid (RA) can activate PPAR-γ, but its effect on cardiac remodeling remains largely unknown. Our study aimed to investigate the effect of RA on cardiac remodeling and to clarify the underlying mechanism. Mice were subjected to aortic banding to generate pressure overload induced cardiac remodeling and then were orally administered RA (100 mg/kg/day) for 7 weeks beginning 1 week after surgery. The morphological examination, echocardiography, and molecular markers were used to evaluate the effects of RA. To ascertain whether the beneficial effect of RA on cardiac fibrosis was mediated by AMPKα, AMPKα2 knockout mice were used. Neonatal rat cardiomyocytes and fibroblasts were separated and cultured to validate the protective effect of RA in vitro. RA-treated mice exhibited a similar hypertrophic response as mice without RA treatment, but had an attenuated fibrotic response and improved cardiac function after pressure overload. Activated AMPKα was essential for the anti-fibrotic effect of RA via inhibiting the phosphorylation and nuclear translocation of Smad3 in vivo and in vitro, and AMPKα deficiency abolished RA-mediated protective effects. Small interfering RNA against Ppar-γ (siPpar-γ) and GW9662, a specific antagonist of PPAR-γ, abolished RA-mediated AMPKα phosphorylation and alleviation of fibrotic response in vitro. RA attenuated cardiac fibrosis following long-term pressure overload via AMPKα/Smad3 signaling and PPAR-γ was required for the activation of AMPKα. RA might be a promising therapeutic agent against cardiac fibrosis.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

Similar articles
-
Piperine Attenuates Pathological Cardiac Fibrosis Via PPAR-γ/AKT Pathways.EBioMedicine. 2017 Apr;18:179-187. doi: 10.1016/j.ebiom.2017.03.021. Epub 2017 Mar 14. EBioMedicine. 2017. PMID: 28330809 Free PMC article.
-
PPAR-γ is involved in the protective effect of 2,3,4',5-tetrahydroxystilbene-2-O-beta-D-glucoside against cardiac fibrosis in pressure-overloaded rats.Eur J Pharmacol. 2016 Nov 15;791:105-114. doi: 10.1016/j.ejphar.2016.08.025. Epub 2016 Aug 26. Eur J Pharmacol. 2016. PMID: 27568841
-
Heat shock transcription factor 1 protects against pressure overload-induced cardiac fibrosis via Smad3.J Mol Med (Berl). 2017 Apr;95(4):445-460. doi: 10.1007/s00109-016-1504-2. Epub 2017 Jan 13. J Mol Med (Berl). 2017. PMID: 28091697 Free PMC article.
-
Metformin attenuates cardiac fibrosis by inhibiting the TGFbeta1-Smad3 signalling pathway.Cardiovasc Res. 2010 Aug 1;87(3):504-13. doi: 10.1093/cvr/cvq066. Epub 2010 Mar 3. Cardiovasc Res. 2010. PMID: 20200042
-
Cartilage intermediate layer protein-1 alleviates pressure overload-induced cardiac fibrosis via interfering TGF-β1 signaling.J Mol Cell Cardiol. 2018 Mar;116:135-144. doi: 10.1016/j.yjmcc.2018.02.006. Epub 2018 Feb 10. J Mol Cell Cardiol. 2018. PMID: 29438665
Cited by
-
MicroRNA-31-5p Exacerbates Lipopolysaccharide-Induced Acute Lung Injury via Inactivating Cab39/AMPKα Pathway.Oxid Med Cell Longev. 2020 Oct 8;2020:8822361. doi: 10.1155/2020/8822361. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 33101593 Free PMC article.
-
miR‑216a exacerbates TGF‑β‑induced myofibroblast transdifferentiation via PTEN/AKT signaling.Mol Med Rep. 2019 Jun;19(6):5345-5352. doi: 10.3892/mmr.2019.10200. Epub 2019 Apr 30. Mol Med Rep. 2019. PMID: 31059054 Free PMC article.
-
Matrine attenuates pathological cardiac fibrosis via RPS5/p38 in mice.Acta Pharmacol Sin. 2021 Apr;42(4):573-584. doi: 10.1038/s41401-020-0473-8. Epub 2020 Jul 21. Acta Pharmacol Sin. 2021. PMID: 32694761 Free PMC article.
-
Taxifolin protects against doxorubicin-induced cardiotoxicity and ferroptosis by adjusting microRNA-200a-mediated Nrf2 signaling pathway.Heliyon. 2023 Nov 8;9(11):e22011. doi: 10.1016/j.heliyon.2023.e22011. eCollection 2023 Nov. Heliyon. 2023. PMID: 38053888 Free PMC article.
-
miR‑351‑5p aggravates lipopolysaccharide‑induced acute lung injury via inhibiting AMPK.Mol Med Rep. 2021 Oct;24(4):689. doi: 10.3892/mmr.2021.12330. Epub 2021 Jul 30. Mol Med Rep. 2021. PMID: 34328196 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

