Mineralized Biomaterials Mediated Repair of Bone Defects Through Endogenous Cells
- PMID: 29368582
- PMCID: PMC6033304
- DOI: 10.1089/ten.TEA.2017.0297
Mineralized Biomaterials Mediated Repair of Bone Defects Through Endogenous Cells
Abstract
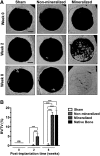
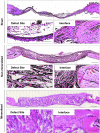
Synthetic biomaterials that create a dynamic calcium (Ca2+)-, phosphate (PO43-) ion-, and calcium phosphate (CaP)-rich microenvironment, similar to that found in native bone tissue, have been shown to promote osteogenic commitment of stem cells in vitro and in vivo. The intrinsic osteoconductivity and osteoinductivity of such biomaterials make them promising bone grafts for the treatment of bone defects. We thus aimed to evaluate the potential of mineralized biomaterials to induce bone repair of a critical-sized cranial defect in the absence of exogenous cells and growth factors. Our results demonstrate that the mineralized biomaterial alone can support complete bone formation within critical-sized bone defects through recruitment of endogenous cells and neo-bone tissue formation in mice. The newly formed bone tissue recapitulated many key characteristics of native bone such as formation of bone minerals reaching similar bone mineral density, presence of bone-forming osteoblasts and tartrate-resistant acid phosphatase-expressing osteoclasts, as well as vascular networks. Biomaterials that recruit endogenous cells and provide a tissue-specific microenvironment to modulate cellular behavior and support generation of functional tissues are a key step forward in moving bench-side tissue engineering approaches to the bedside. Such tissue engineering strategies could eventually pave the path toward readily available therapies that significantly reduce patient cost of care and improve overall clinical outcomes.
Keywords: bone repair; critical defect; mineralized biomaterial; osteoinductive.
Conflict of interest statement
No competing financial interests exist.
Figures



References
-
- Giannoudis P.V., Dinopoulos H., and Tsiridis E. Bone substitutes: an update. Injury 36, 20, 2005 - PubMed
-
- Lichte P., Pape H.C., Pufe T., Kobbe P., and Fischer H. Scaffolds for bone healing: concepts, materials and evidence. Injury 42, 569, 2011 - PubMed
-
- Laurie S.W.S., Kaban L.B., Mulliken J.B., and Murray J.E. Donor-site morbidity after harvesting rib and iliac bone. Plast Reconstr Surg 73, 933, 1984 - PubMed
-
- Zlotolow D.A., Vaccaro A.R., Salamon M.L., and Albert T.J. The role of human bone morphogenetic proteins in spinal fusion. J Am Acad Orthop Surg 8, 3, 2000 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

