Impact of EML4-ALK Variant on Resistance Mechanisms and Clinical Outcomes in ALK-Positive Lung Cancer
- PMID: 29373100
- PMCID: PMC5903999
- DOI: 10.1200/JCO.2017.76.2294
Impact of EML4-ALK Variant on Resistance Mechanisms and Clinical Outcomes in ALK-Positive Lung Cancer
Abstract
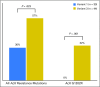
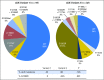
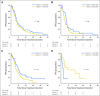
Purpose Advanced anaplastic lymphoma kinase ( ALK) fusion-positive non-small-cell lung cancers (NSCLCs) are effectively treated with ALK tyrosine kinase inhibitors (TKIs). However, clinical outcomes in these patients vary, and the benefit of TKIs is limited as a result of acquired resistance. Emerging data suggest that the ALK fusion variant may affect clinical outcome, but the molecular basis for this association is unknown. Patients and Methods We identified 129 patients with ALK-positive NSCLC with known ALK variants. ALK resistance mutations and clinical outcomes on ALK TKIs were retrospectively evaluated according to ALK variant. A Foundation Medicine data set of 577 patients with ALK-positive NSCLC was also examined. Results The most frequent ALK variants were EML4-ALK variant 1 in 55 patients (43%) and variant 3 in 51 patients (40%). We analyzed 77 tumor biopsy specimens from patients with variants 1 and 3 who had progressed on an ALK TKI. ALK resistance mutations were significantly more common in variant 3 than in variant 1 (57% v 30%; P = .023). In particular, ALK G1202R was more common in variant 3 than in variant 1 (32% v 0%; P < .001). Analysis of the Foundation Medicine database revealed similar associations of variant 3 with ALK resistance mutation and with G1202R ( P = .010 and .015, respectively). Among patients treated with the third-generation ALK TKI lorlatinib, variant 3 was associated with a significantly longer progression-free survival than variant 1 (hazard ratio, 0.31; 95% CI, 0.12 to 0.79; P = .011). Conclusion Specific ALK variants may be associated with the development of ALK resistance mutations, particularly G1202R, and provide a molecular link between variant and clinical outcome. ALK variant thus represents a potentially important factor in the selection of next-generation ALK inhibitors.
Figures


Comment in
-
EML4-ALK Variant Affects ALK Resistance Mutations.J Clin Oncol. 2018 Apr 20;36(12):1257-1259. doi: 10.1200/JCO.2017.77.2764. Epub 2018 Mar 1. J Clin Oncol. 2018. PMID: 29494260 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

