Increased Exposure of Tacrolimus by Co-administered Mycophenolate Mofetil: Population Pharmacokinetic Analysis in Healthy Volunteers
- PMID: 29374217
- PMCID: PMC5786104
- DOI: 10.1038/s41598-018-20071-3
Increased Exposure of Tacrolimus by Co-administered Mycophenolate Mofetil: Population Pharmacokinetic Analysis in Healthy Volunteers
Abstract
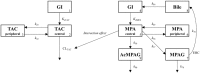
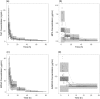
The objective of the study was to investigate the pharmacokinetic drug-drug interactions between tacrolimus (TAC) and mycophenolate mofetil (MMF) in healthy Korean male volunteers. Seventeen volunteers participated in a three-period, single-dose, and fixed sequence study. They sequentially received MMF, TAC, and the combination. Concentrations of TAC, mycophenolic acid (MPA), and its metabolites MPA 7-O-glucuronide and MPA acyl glucuronide were measured. The variants of CYP3A4, CYP3A5, SLCO1B1, SLCO1B3, ABCC2, UGT1A9, and UGT2B7 were genotyped. Drug interaction was evaluated with a non-compartmental analysis and population pharmacokinetic modelling to quantify the interaction effect. A total of 1,082 concentrations of those analytes were analysed. AUC0-inf of TAC increased by 22.1% (322.4 ± 174.1 to 393.6 ± 121.7 ng·h/mL; P < 0.05) when co-administered with MMF, whereas the pharmacokinetic parameters of MPA and its metabolites were not changed by TAC. Apparent clearance (CL/F) of TAC was 17.8 L/h [relative standard error (RSE) 11%] or 13.8 L/h (RSE 11%) without or with MMF, respectively. Interaction was explained by the exponential model. The CYP3A5 genotype was the only significant covariate. The population estimate of CL/F of TAC was 1.48-fold (RSE 16%) in CYP3A5 expressers when compared to nonexpressers. CL/F of TAC was decreased when co-administered with MMF in these subjects.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Model based development of tacrolimus dosing algorithm considering CYP3A5 genotypes and mycophenolate mofetil drug interaction in stable kidney transplant recipients.Sci Rep. 2019 Aug 13;9(1):11740. doi: 10.1038/s41598-019-47876-0. Sci Rep. 2019. PMID: 31409869 Free PMC article.
-
Genetic polymorphisms in metabolic enzymes and transporters have no impact on mycophenolic acid pharmacokinetics in adult kidney transplant patients co-treated with tacrolimus: A population analysis.J Clin Pharm Ther. 2021 Dec;46(6):1564-1575. doi: 10.1111/jcpt.13488. Epub 2021 Jul 26. J Clin Pharm Ther. 2021. PMID: 34312870
-
Pharmacokinetics, efficacy, and safety of mycophenolate mofetil in combination with standard-dose or reduced-dose tacrolimus in liver transplant recipients.Liver Transpl. 2009 Feb;15(2):136-47. doi: 10.1002/lt.21657. Liver Transpl. 2009. PMID: 19177449 Clinical Trial.
-
Clinical pharmacokinetics of mycophenolate mofetil.Clin Pharmacokinet. 1998 Jun;34(6):429-55. doi: 10.2165/00003088-199834060-00002. Clin Pharmacokinet. 1998. PMID: 9646007 Review.
-
Pharmacokinetics of mycophenolate mofetil and sirolimus in children.Ther Drug Monit. 2008 Apr;30(2):138-42. doi: 10.1097/FTD.0b013e31816ba73a. Ther Drug Monit. 2008. PMID: 18367972 Review.
Cited by
-
Association of ABCC2 Haplotypes to Mycophenolic Acid Pharmacokinetics in Stable Kidney Transplant Recipients.J Clin Pharmacol. 2021 Dec;61(12):1592-1605. doi: 10.1002/jcph.1932. Epub 2021 Jul 20. J Clin Pharmacol. 2021. PMID: 34169529 Free PMC article.
-
Impact of the CYP3A5*1 Allele on the Pharmacokinetics of Tacrolimus in Japanese Heart Transplant Patients.Eur J Drug Metab Pharmacokinet. 2018 Dec;43(6):665-673. doi: 10.1007/s13318-018-0478-6. Eur J Drug Metab Pharmacokinet. 2018. PMID: 29691732
-
Toward a robust tool for pharmacokinetic-based personalization of treatment with tacrolimus in solid organ transplantation: A model-based meta-analysis approach.Br J Clin Pharmacol. 2019 Dec;85(12):2793-2823. doi: 10.1111/bcp.14110. Epub 2019 Dec 17. Br J Clin Pharmacol. 2019. PMID: 31471970 Free PMC article.
-
Influence of Calcineurin Inhibitor and Sex on Mycophenolic Acid Pharmacokinetics and Adverse Effects Post-Renal Transplant.J Clin Pharmacol. 2019 Oct;59(10):1351-1365. doi: 10.1002/jcph.1428. Epub 2019 May 6. J Clin Pharmacol. 2019. PMID: 31062373 Free PMC article.
-
A preliminary exploration of liver microsomes and PBPK to uncover the impact of CYP3A4/5 and CYP2C19 on tacrolimus and voriconazole drug-drug interactions.Sci Rep. 2025 Feb 21;15(1):6389. doi: 10.1038/s41598-025-91356-7. Sci Rep. 2025. PMID: 39984708 Free PMC article.
References
-
- Johnson C, et al. Randomized trial of tacrolimus (Prograf) in combination with azathioprine or mycophenolate mofetil versus cyclosporine (Neoral) with mycophenolate mofetil after cadaveric kidney transplantation. Transplantation. 2000;69:834–841. doi: 10.1097/00007890-200003150-00028. - DOI - PubMed
-
- Kidney Disease: Improving Global Outcomes Transplant Work, G. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplant9Suppl 3, S1–155 10.1111/j.1600-6143.2009.02834.x (2009). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

