The Beta Adrenergic Receptor Blocker Propranolol Counteracts Retinal Dysfunction in a Mouse Model of Oxygen Induced Retinopathy: Restoring the Balance between Apoptosis and Autophagy
- PMID: 29375312
- PMCID: PMC5770647
- DOI: 10.3389/fncel.2017.00395
The Beta Adrenergic Receptor Blocker Propranolol Counteracts Retinal Dysfunction in a Mouse Model of Oxygen Induced Retinopathy: Restoring the Balance between Apoptosis and Autophagy
Abstract
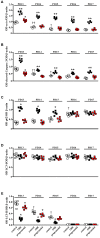
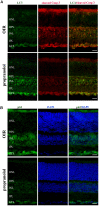
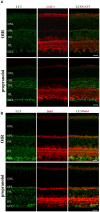
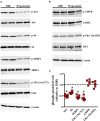
In a mouse model of oxygen induced retinopathy (OIR), beta adrenergic receptor (BAR) blockade has been shown to recover hypoxia-associated retinal damages. Although the adrenergic signaling is an important regulator of apoptotic and autophagic processes, the role of BARs in retinal cell death remains to be elucidated. The present study was aimed at investigating whether ameliorative effects of BAR blockers may occur through their coordinated action on apoptosis and autophagy. To this aim, retinas from control and OIR mice untreated or treated with propranolol, a non-selective BAR1/2 blocker, were characterized in terms of expression and localization of apoptosis and autophagy markers. The effects of propranolol on autophagy signaling were also evaluated and specific autophagy modulators were used to get functional information on the autophagic effects of BAR antagonism. Finally, propranolol effects on neurodegenerative processes were associated to an electrophysiological investigation of retinal function by recording electroretinogram (ERG). We found that retinas of OIR mice are characterized by increased apoptosis and decreased autophagy, while propranolol reduces apoptosis and stimulates autophagy. In particular, propranolol triggers autophagosome formation in bipolar, amacrine and ganglion cells that are committed to die by apoptosis in response to hypoxia. Also our data argue that propranolol, through the inhibition of the Akt-mammalian target of rapamycin pathway, activates autophagy which decreases retinal cell death. At the functional level, propranolol recovers dysfunctional ERG by recovering the amplitude of a- and b-waves, and oscillatory potentials, thus indicating an efficient restoring of retinal transduction. Overall, our results demonstrate that BAR1/2 are key regulators of retinal apoptosis/autophagy, and that BAR1/2 blockade leads to autophagy-mediated neuroprotection. Reinstating the balance between apoptotic and autophagic machines may therefore be viewed as a future goal in the treatment of retinopathies.
Keywords: apoptosis; autophagy; beta adrenergic receptors; electroretinogram; proliferative retinopathies; propranolol; retinal neurons.
Figures





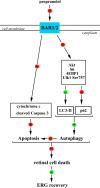
Similar articles
-
Effect of Autophagy Modulators on Vascular, Glial, and Neuronal Alterations in the Oxygen-Induced Retinopathy Mouse Model.Front Cell Neurosci. 2019 Jun 26;13:279. doi: 10.3389/fncel.2019.00279. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31297049 Free PMC article.
-
Role of the adrenergic system in a mouse model of oxygen-induced retinopathy: antiangiogenic effects of beta-adrenoreceptor blockade.Invest Ophthalmol Vis Sci. 2011 Jan 5;52(1):155-70. doi: 10.1167/iovs.10-5536. Invest Ophthalmol Vis Sci. 2011. PMID: 20739470
-
Antiangiogenic effects of β2 -adrenergic receptor blockade in a mouse model of oxygen-induced retinopathy.J Neurochem. 2011 Dec;119(6):1317-29. doi: 10.1111/j.1471-4159.2011.07530.x. Epub 2011 Nov 2. J Neurochem. 2011. PMID: 21988318
-
The β-adrenergic system as a possible new target for pharmacologic treatment of neovascular retinal diseases.Prog Retin Eye Res. 2014 Sep;42:103-29. doi: 10.1016/j.preteyeres.2014.06.001. Epub 2014 Jun 14. Prog Retin Eye Res. 2014. PMID: 24933041 Review.
-
Infantile hemangiomas, retinopathy of prematurity and cancer: a common pathogenetic role of the β-adrenergic system.Med Res Rev. 2015 May;35(3):619-52. doi: 10.1002/med.21336. Epub 2014 Dec 19. Med Res Rev. 2015. PMID: 25523517 Review.
Cited by
-
Decoupling Oxygen Tension From Retinal Vascularization as a New Perspective for Management of Retinopathy of Prematurity. New Opportunities From β-adrenoceptors.Front Pharmacol. 2022 Jan 21;13:835771. doi: 10.3389/fphar.2022.835771. eCollection 2022. Front Pharmacol. 2022. PMID: 35126166 Free PMC article. Review.
-
Agonism of β3-Adrenoceptors Inhibits Pathological Retinal Angiogenesis in the Model of Oxygen-Induced Retinopathy.Invest Ophthalmol Vis Sci. 2024 Aug 1;65(10):34. doi: 10.1167/iovs.65.10.34. Invest Ophthalmol Vis Sci. 2024. PMID: 39186263 Free PMC article.
-
Novel Insights into Beta 2 Adrenergic Receptor Function in the rd10 Model of Retinitis Pigmentosa.Cells. 2020 Sep 9;9(9):2060. doi: 10.3390/cells9092060. Cells. 2020. PMID: 32917020 Free PMC article.
-
Propranolol: a new pharmacologic approach to counter retinopathy of prematurity progression.Front Pediatr. 2024 Jan 16;12:1322783. doi: 10.3389/fped.2024.1322783. eCollection 2024. Front Pediatr. 2024. PMID: 38292211 Free PMC article. Review.
-
Dysfunctional autophagy induced by the pro-apoptotic natural compound climacostol in tumour cells.Cell Death Dis. 2018 Dec 19;10(1):10. doi: 10.1038/s41419-018-1254-x. Cell Death Dis. 2018. PMID: 30584259 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

