Identification and Characterization of 5' Untranslated Regions (5'UTRs) in Zymomonas mobilis as Regulatory Biological Parts
- PMID: 29375488
- PMCID: PMC5770649
- DOI: 10.3389/fmicb.2017.02432
Identification and Characterization of 5' Untranslated Regions (5'UTRs) in Zymomonas mobilis as Regulatory Biological Parts
Abstract
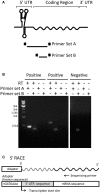
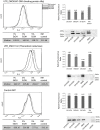
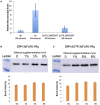
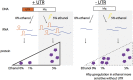
Regulatory RNA regions within a transcript, particularly in the 5' untranslated region (5'UTR), have been shown in a variety of organisms to control the expression levels of these mRNAs in response to various metabolites or environmental conditions. Considering the unique tolerance of Zymomonas mobilis to ethanol and the growing interest in engineering microbial strains with enhanced tolerance to industrial inhibitors, we searched natural cis-regulatory regions in this microorganism using transcriptomic data and bioinformatics analysis. Potential regulatory 5'UTRs were identified and filtered based on length, gene function, relative gene counts, and conservation in other organisms. An in vivo fluorescence-based screening system was developed to confirm the responsiveness of 36 5'UTR candidates to ethanol, acetate, and xylose stresses. UTR_ZMO0347 (5'UTR of gene ZMO0347 encoding the RNA binding protein Hfq) was found to down-regulate downstream gene expression under ethanol stress. Genomic deletion of UTR_ZMO0347 led to a general decrease of hfq expression at the transcript level and increased sensitivity for observed changes in Hfq expression at the protein level. The role of UTR_ZMO0347 and other 5'UTRs gives us insight into the regulatory network of Z. mobilis in response to stress and unlocks new strategies for engineering robust industrial strains as well as for harvesting novel responsive regulatory biological parts for controllable gene expression platforms in this organism.
Keywords: 5′ UTR; 5′ rapid amplification of cDNA ends (RACE); Zymomonas mobilis; regulatory RNA; stress response.
Figures


Similar articles
-
The Zymomonas mobilis regulator hfq contributes to tolerance against multiple lignocellulosic pretreatment inhibitors.BMC Microbiol. 2010 May 7;10:135. doi: 10.1186/1471-2180-10-135. BMC Microbiol. 2010. PMID: 20459639 Free PMC article.
-
Molecular mechanism of enhanced ethanol tolerance associated with hfq overexpression in Zymomonas mobilis.Front Bioeng Biotechnol. 2022 Dec 15;10:1098021. doi: 10.3389/fbioe.2022.1098021. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36588936 Free PMC article.
-
Growth hormone (GH) receptor and GH-binding protein messenger ribonucleic acids with alternative 5'-untranslated regions are differentially expressed in mouse liver and placenta.Endocrinology. 1995 Jul;136(7):2913-21. doi: 10.1210/endo.136.7.7789316. Endocrinology. 1995. PMID: 7789316
-
Zymomonas mobilis--science and industrial application.Crit Rev Biotechnol. 1993;13(1):57-98. doi: 10.3109/07388559309069198. Crit Rev Biotechnol. 1993. PMID: 8477453 Review.
-
New technologies provide more metabolic engineering strategies for bioethanol production in Zymomonas mobilis.Appl Microbiol Biotechnol. 2019 Mar;103(5):2087-2099. doi: 10.1007/s00253-019-09620-6. Epub 2019 Jan 19. Appl Microbiol Biotechnol. 2019. PMID: 30661108 Review.
Cited by
-
Design and construction of microbial cell factories based on systems biology.Synth Syst Biotechnol. 2022 Nov 18;8(1):176-185. doi: 10.1016/j.synbio.2022.11.001. eCollection 2023 Mar. Synth Syst Biotechnol. 2022. PMID: 36874510 Free PMC article. Review.
-
Improved GFP Variants to Study Gene Expression in Haloarchaea.Front Microbiol. 2019 May 29;10:1200. doi: 10.3389/fmicb.2019.01200. eCollection 2019. Front Microbiol. 2019. PMID: 31191505 Free PMC article.
-
Prediction and characterization of promoters and ribosomal binding sites of Zymomonas mobilis in system biology era.Biotechnol Biofuels. 2019 Mar 14;12:52. doi: 10.1186/s13068-019-1399-6. eCollection 2019. Biotechnol Biofuels. 2019. PMID: 30911332 Free PMC article.
-
Genome-Scale Transcription-Translation Mapping Reveals Features of Zymomonas mobilis Transcription Units and Promoters.mSystems. 2020 Jul 21;5(4):e00250-20. doi: 10.1128/mSystems.00250-20. mSystems. 2020. PMID: 32694125 Free PMC article.
-
Comprehensive network of stress-induced responses in Zymomonas mobilis during bioethanol production: from physiological and molecular responses to the effects of system metabolic engineering.Microb Cell Fact. 2024 Jun 18;23(1):180. doi: 10.1186/s12934-024-02459-1. Microb Cell Fact. 2024. PMID: 38890644 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

