Carcinoma associated fibroblasts derived from oral squamous cell carcinoma promote lymphangiogenesis via c-Met/PI3K/AKT in vitro
- PMID: 29375714
- PMCID: PMC5766077
- DOI: 10.3892/ol.2017.7301
Carcinoma associated fibroblasts derived from oral squamous cell carcinoma promote lymphangiogenesis via c-Met/PI3K/AKT in vitro
Abstract
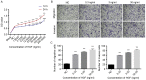
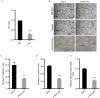
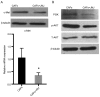
Carcinoma-associated fibroblasts (CAFs) are dominant components of the tumor microenvironment (TME) that promote the development, progression and metastasis of cancer. c-Met is a receptor of the hepatocyte growth factor (HGF), which is involved in lymphangiogenesis. Currently, the roles of CAFs during lymphangiogenesis are unknown. It has been hypothesized that CAFs contribute to lymphangiogenesis of oral squamous cell carcinoma (OSCC) via a HGF/c-Met complex. The expression of HGF in OSCC was determined using CAFs derived from OSCC tissue and it was demonstrated that HGF is overexpressed in OSCC-derived CAFs. It was also revealed that c-Met was highly expressed in human lymphatic endothelial cells (HLECs) when co-cultured with CAFs. Furthermore, it was demonstrated that recombinant human HGF significantly enhanced the proliferation, migration, invasion and tube formation of HLECs. By contrast, the inhibition of c-Met expression suppressed the aforementioned biological activities and also downregulated the expression of c-Met, phosphoinositide 3-kinase and phosphorylated protein kinase B. Taken together, these data demonstrate that c-Met is associated with the regulation of lymphangiogenesis. Thus, the results of the present study indicate that c-Met may be a promising novel therapeutic target to treat patients with OSCC.
Keywords: c-Met; carcinoma-associated fibroblasts; hepatocyte growth factor; lymphangiogenesis; oral squamous cell carcinoma.
Figures


Similar articles
-
Significance of oral cancer-associated fibroblasts in angiogenesis, lymphangiogenesis, and tumor invasion in oral squamous cell carcinoma.J Oral Pathol Med. 2017 Jan;46(1):21-30. doi: 10.1111/jop.12452. Epub 2016 May 27. J Oral Pathol Med. 2017. PMID: 27229731
-
CAF-derived HGF promotes cell proliferation and drug resistance by up-regulating the c-Met/PI3K/Akt and GRP78 signalling in ovarian cancer cells.Biosci Rep. 2017 Apr 10;37(2):BSR20160470. doi: 10.1042/BSR20160470. Print 2017 Apr 28. Biosci Rep. 2017. PMID: 28258248 Free PMC article.
-
An HGF-dependent positive feedback loop between bladder cancer cells and fibroblasts mediates lymphangiogenesis and lymphatic metastasis.Cancer Commun (Lond). 2023 Dec;43(12):1289-1311. doi: 10.1002/cac2.12470. Epub 2023 Jul 22. Cancer Commun (Lond). 2023. PMID: 37483113 Free PMC article.
-
Roles of prostaglandins in tumor-associated lymphangiogenesis with special reference to breast cancer.Cancer Metastasis Rev. 2018 Sep;37(2-3):369-384. doi: 10.1007/s10555-018-9734-0. Cancer Metastasis Rev. 2018. PMID: 29858743 Review.
-
Roles of Mitochondria in Oral Squamous Cell Carcinoma Therapy: Friend or Foe?Cancers (Basel). 2022 Nov 22;14(23):5723. doi: 10.3390/cancers14235723. Cancers (Basel). 2022. PMID: 36497206 Free PMC article. Review.
Cited by
-
Lymphatic vasculature in tumor metastasis and immunobiology.J Zhejiang Univ Sci B. 2019 Jul 12;21(1):3-11. doi: 10.1631/jzus.B1800633. Online ahead of print. J Zhejiang Univ Sci B. 2019. PMID: 31317681 Free PMC article. Review.
-
Desmoplastic reaction in the microenvironment of head and neck and other solid tumors: the therapeutic barrier.Ther Adv Med Oncol. 2025 Feb 7;17:17588359251317144. doi: 10.1177/17588359251317144. eCollection 2025. Ther Adv Med Oncol. 2025. PMID: 39926258 Free PMC article. Review.
-
The diverse role of oral fibroblasts in normal and disease.J Oral Maxillofac Pathol. 2022 Jan-Mar;26(1):6-13. doi: 10.4103/jomfp.jomfp_48_22. Epub 2022 Mar 31. J Oral Maxillofac Pathol. 2022. PMID: 35571294 Free PMC article. Review.
-
Role of lymphatic endothelial cells in the tumor microenvironment-a narrative review of recent advances.Transl Lung Cancer Res. 2021 May;10(5):2252-2277. doi: 10.21037/tlcr-21-40. Transl Lung Cancer Res. 2021. PMID: 34164274 Free PMC article. Review.
-
Human colorectal cancer-derived carcinoma associated fibroblasts promote CD44-mediated adhesion of colorectal cancer cells to endothelial cells by secretion of HGF.Cancer Cell Int. 2019 Jul 24;19:192. doi: 10.1186/s12935-019-0914-y. eCollection 2019. Cancer Cell Int. 2019. PMID: 31367190 Free PMC article.
References
-
- Roberts TJ, Colevas AD, Hara W, Holsinger FC, Oakley-Girvan I, Divi V. Number of positive nodes is superior to the lymph node ratio and American Joint Committee on Cancer N staging for the prognosis of surgically treated head and neck squamous cell carcinomas. Cancer. 2016;122:1388–1397. doi: 10.1002/cncr.29932. - DOI - PubMed
-
- Fullár A, Kovalszky I, Bitsche M, Romani A, Schartinger VH, Sprinzl GM, Riechelmann H, Dudás J. Tumor cell and carcinoma-associated fibroblast interaction regulates matrix metalloproteinases and their inhibitors in oral squamous cell carcinoma. Exp Cell Res. 2012;318:1517–1527. doi: 10.1016/j.yexcr.2012.03.023. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
