Severe Toxicity in Nonhuman Primates and Piglets Following High-Dose Intravenous Administration of an Adeno-Associated Virus Vector Expressing Human SMN
- PMID: 29378426
- PMCID: PMC5865262
- DOI: 10.1089/hum.2018.015
Severe Toxicity in Nonhuman Primates and Piglets Following High-Dose Intravenous Administration of an Adeno-Associated Virus Vector Expressing Human SMN
Abstract
Neurotropic adeno-associated virus (AAV) serotypes such as AAV9 have been demonstrated to transduce spinal alpha motor neurons when administered intravenously (i.v.) at high doses. This observation led to the recent successful application of i.v. AAV9 delivery to treat infants with spinal muscular atrophy, an inherited deficiency of the survival of motor neuron (SMN) protein characterized by selective death of lower motor neurons. To evaluate the efficiency of motor neuron transduction with an AAV9 variant (AAVhu68) using this approach, three juvenile nonhuman primates (NHPs; aged 14 months) and three piglets (aged 7-30 days) were treated with an i.v. injection of an AAVhu68 vector carrying a human SMN transgene at a dose similar to that employed in the spinal muscular atrophy clinical trial. Administration of 2 × 1014 genome copies per kilogram of body weight resulted in widespread transduction of spinal motor neurons in both species. However, severe toxicity occurred in both NHPs and piglets. All three NHPs exhibited marked transaminase elevations. In two NHPs, the transaminase elevations resolved without clinical sequelae, while one NHP developed acute liver failure and shock and was euthanized 4 days after vector injection. Degeneration of dorsal root ganglia sensory neurons was also observed, although NHPs exhibited no clinically apparent sensory deficits. There was no correlation between clinical findings and T-cell responses to the vector capsid or transgene product in NHPs. Piglets demonstrated no evidence of hepatic toxicity, but within 14 days of vector injection, all three animals exhibited proprioceptive deficits and ataxia, which profoundly impaired ambulation and necessitated euthanasia. These clinical findings correlated with more severe dorsal root ganglia sensory neuron lesions than those observed in NHPs. The liver and sensory neuron findings appear to be a direct consequence of AAV transduction independent of an immune response to the capsid or transgene product. The present results and those of another recent study utilizing a different AAV9 variant and transgene indicate that systemic and sensory neuron toxicity may be general properties of i.v. delivery of AAV vectors at high doses, irrespective of the capsid serotype or transgene. Preclinical and clinical studies involving high systemic doses of AAV vectors should include careful monitoring for similar toxicities.
Keywords: adeno-associated virus; axonopathy; gene therapy; hepatic toxicity; liver failure; shock.
Conflict of interest statement
J.M.W. is an advisor to, a founder of, holds equity in, and has a sponsored research agreement with REGENXBIO. He is a former consultant to and holds stock in Solid Biosciences. He also has a sponsored research agreement with Ultragenyx. In addition, he is a consultant to several biopharmaceutical companies. He is also an inventor on patents licensed to various biopharmaceutical companies. The remaining authors declare no potential competing financial interests.
Figures

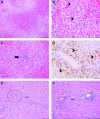



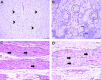
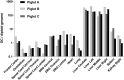

Comment in
-
Safety First: Perspective on Patient-Centered Development of AAV Gene Therapy Products.Mol Ther. 2018 Mar 7;26(3):669-671. doi: 10.1016/j.ymthe.2018.02.009. Epub 2018 Mar 1. Mol Ther. 2018. PMID: 29503193 Free PMC article. No abstract available.
References
-
- Lu YY, Wang LJ, Muramatsu S, et al. . Intramuscular injection of AAV-GDNF results in sustained expression of transgenic GDNF, and its delivery to spinal motoneurons by retrograde transport. Neurosci Res 2003;45:33–40 - PubMed
-
- Hollis Ii ER, Kadoya K, Hirsch M, et al. . Efficient retrograde neuronal transduction utilizing self-complementary AAV1. Mol Ther 2008;16:296–301 - PubMed
-
- Kaspar BK, Lladó J, Sherkat N, et al. . Retrograde viral delivery of IGF-1 prolongs survival in a mouse ALS model. Science 2003;301:839–842 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

