Dual catalysis for enantioselective convergent synthesis of enantiopure vicinal amino alcohols
- PMID: 29379007
- PMCID: PMC5789022
- DOI: 10.1038/s41467-017-02698-4
Dual catalysis for enantioselective convergent synthesis of enantiopure vicinal amino alcohols
Abstract
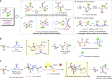
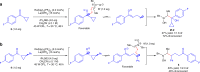
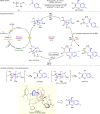
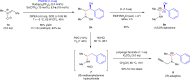
Enantiopure vicinal amino alcohols and derivatives are essential structural motifs in natural products and pharmaceutically active molecules, and serve as main chiral sources in asymmetric synthesis. Currently known asymmetric catalytic protocols for this class of compounds are still rare and often suffer from limited scope of substrates, relatively low regio- or stereoselectivities, thus prompting the development of more effective methodologies. Herein we report a dual catalytic strategy for the convergent enantioselective synthesis of vicinal amino alcohols. The method features a radical-type Zimmerman-Traxler transition state formed from a rare earth metal with a nitrone and an aromatic ketyl radical in the presence of chiral N,N'-dioxide ligands. In addition to high level of enantio- and diastereoselectivities, our synthetic protocol affords advantages of simple operation, mild conditions, high-yielding, and a broad scope of substrates. Furthermore, this protocol has been successfully applied to the concise synthesis of pharmaceutically valuable compounds (e.g., ephedrine and selegiline).
Conflict of interest statement
The authors declare no competing financial interests.
Figures
Similar articles
-
An advance on exploring N-tert-butanesulfinyl imines in asymmetric synthesis of chiral amines.Acc Chem Res. 2008 Jul;41(7):831-40. doi: 10.1021/ar7002623. Epub 2008 Jun 6. Acc Chem Res. 2008. PMID: 18533688
-
Asymmetric reduction of α-amino ketones with a KBH4 solution catalyzed by chiral Lewis acids.Chemistry. 2014 Oct 13;20(42):13482-6. doi: 10.1002/chem.201404732. Epub 2014 Sep 15. Chemistry. 2014. PMID: 25223259
-
Photochemical Stereocontrol Using Tandem Photoredox-Chiral Lewis Acid Catalysis.Acc Chem Res. 2016 Oct 18;49(10):2307-2315. doi: 10.1021/acs.accounts.6b00280. Epub 2016 Aug 9. Acc Chem Res. 2016. PMID: 27505691 Free PMC article.
-
Enzymatic asymmetric synthesis of chiral amino acids.Chem Soc Rev. 2018 Feb 19;47(4):1516-1561. doi: 10.1039/c7cs00253j. Chem Soc Rev. 2018. PMID: 29362736 Review.
-
[Synthesis and application of novel biaryl compounds with axial chirality as catalysts in enantioselective reactions].Yakugaku Zasshi. 2000 Jan;120(1):68-75. doi: 10.1248/yakushi1947.120.1_68. Yakugaku Zasshi. 2000. PMID: 10655783 Review. Japanese.
Cited by
-
Bis-Cyclometalated Indazole Chiral-at-Rhodium Catalyst for Asymmetric Photoredox Cyanoalkylations.Chemistry. 2019 Dec 2;25(67):15333-15340. doi: 10.1002/chem.201903369. Epub 2019 Nov 13. Chemistry. 2019. PMID: 31541505 Free PMC article.
-
Recent advances in the chemistry of ketyl radicals.Chem Soc Rev. 2021 May 7;50(9):5349-5365. doi: 10.1039/d0cs00358a. Epub 2021 Mar 23. Chem Soc Rev. 2021. PMID: 33972956 Free PMC article.
-
Synthesis of anti-depressant molecules via metal-catalyzed reactions: a review.RSC Adv. 2024 Feb 26;14(10):6948-6971. doi: 10.1039/d3ra06391g. eCollection 2024 Feb 21. RSC Adv. 2024. PMID: 38410364 Free PMC article. Review.
-
Nickel-Catalyzed Defluorinative Coupling of Aliphatic Aldehydes with Trifluoromethyl Alkenes.ACS Catal. 2022 Feb 18;12(4):2463-2471. doi: 10.1021/acscatal.1c05801. Epub 2022 Feb 3. ACS Catal. 2022. PMID: 35992737 Free PMC article.
-
NickelII-catalyzed asymmetric photoenolization/Mannich reaction of (2-alkylphenyl) ketones.Chem Sci. 2022 Jun 22;13(29):8576-8582. doi: 10.1039/d2sc02721f. eCollection 2022 Jul 29. Chem Sci. 2022. PMID: 35974747 Free PMC article.
References
-
- Bergmeier SC. The synthesis of vicinal amino alcohols. Tetrahedron. 2000;56:2561–2576. doi: 10.1016/S0040-4020(00)00149-6. - DOI
-
- Weng C, Zhang H, Xiong X, Lu X, Zhou Y. Evolution of epoxides to synthesize β-amino alcohols. Asian J. Chem. 2014;26:3761–3768.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

