Racial disparities in the rate of cardiotoxicity of HER2-targeted therapies among women with early breast cancer
- PMID: 29381193
- PMCID: PMC5910274
- DOI: 10.1002/cncr.31260
Racial disparities in the rate of cardiotoxicity of HER2-targeted therapies among women with early breast cancer
Abstract
Background: Human epidermal growth factor receptor 2 (HER2)-targeted therapies are highly effective at preventing breast cancer recurrence but are associated with cardiotoxicity in some patients, and minimal data are available regarding racial disparities in the incidence of this toxicity. The authors conducted a retrospective study to analyze the association of black or white race with treatment-induced cardiotoxicity and incomplete therapy among patients with HER2-positive early breast cancer.
Methods: Women with HER2-positive, stage I through III breast cancer who initiated (neo)adjuvant HER2-targeted therapy (trastuzumab with or without pertuzumab) from January 2005 to March 2015 at the authors' institution were eligible. We analyzed differences in the incidence of cardiotoxicity (a decline in the left ventricular ejection fraction to <50% AND an absolute drop in the left ventricular ejection fraction of ≥10% from baseline) and incomplete therapy (<52 weeks of HER2-targeted therapy) between black and white women in univariate and multivariable analyses.
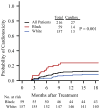
Results: The authors identified 59 black patients and 157 white patients who had a median follow-up 5.2 years. The median patient age was 53 years and was similar for black and white patients. The 1-year cardiotoxicity incidence was 12% overall (95% confidence interval [CI], 7%-16%), 24% in black women (95% CI, 12%-34%), and 7% in white women (95% CI, 3%-11%). Black patients had a significantly greater probability of incomplete therapy compared with white patients (odds ratio, 4.61; 95% CI, 1.70-13.07; P = .002). High correlation was observed between a cardiotoxicity event and incomplete therapy (96% concordance).
Conclusions: Black patients have a higher rate of cardiotoxicity and resultant incomplete adjuvant HER2-targeted therapy than white patients. This patient population may benefit from enhanced cardiac surveillance, cardioprotective strategies, and early referral to cardiology when appropriate. Cancer 2018;124:1904-11. © 2018 American Cancer Society.
Keywords: breast cancer; cardiotoxicity; human epidermal growth factor receptor 2 (HER2)-targeted therapy; pertuzumab; race; trastuzumab.
© 2018 American Cancer Society.
Conflict of interest statement
Figures
Similar articles
-
Racial and Socioeconomic Disparities in Cardiotoxicity Among Women With HER2-Positive Breast Cancer.Am J Cardiol. 2021 May 15;147:116-121. doi: 10.1016/j.amjcard.2021.02.013. Epub 2021 Feb 20. Am J Cardiol. 2021. PMID: 33617819 Free PMC article.
-
Cardiac Safety of Dual Anti-HER2 Therapy in the Neoadjuvant Setting for Treatment of HER2-Positive Breast Cancer.Oncologist. 2017 Jun;22(6):642-647. doi: 10.1634/theoncologist.2016-0406. Epub 2017 Mar 24. Oncologist. 2017. PMID: 28341761 Free PMC article.
-
Pertuzumab, trastuzumab, and standard anthracycline- and taxane-based chemotherapy for the neoadjuvant treatment of patients with HER2-positive localized breast cancer (BERENICE): a phase II, open-label, multicenter, multinational cardiac safety study.Ann Oncol. 2018 Mar 1;29(3):646-653. doi: 10.1093/annonc/mdx773. Ann Oncol. 2018. PMID: 29253081 Free PMC article. Clinical Trial.
-
One year versus a shorter duration of adjuvant trastuzumab for HER2-positive early breast cancer: a systematic review and meta-analysis.Breast Cancer Res Treat. 2019 Jan;173(2):247-254. doi: 10.1007/s10549-018-5001-x. Epub 2018 Oct 13. Breast Cancer Res Treat. 2019. PMID: 30317424
-
Adjuvant trastuzumab regimen for HER2-positive early-stage breast cancer: a systematic review and meta-analysis.Expert Rev Clin Pharmacol. 2019 Aug;12(8):815-824. doi: 10.1080/17512433.2019.1637252. Epub 2019 Jul 9. Expert Rev Clin Pharmacol. 2019. PMID: 31287333 Free PMC article.
Cited by
-
Cardiovascular Considerations Before Cancer Therapy: Gaps in Evidence and JACC: CardioOncology Expert Panel Recommendations.JACC CardioOncol. 2024 Sep 24;6(5):631-654. doi: 10.1016/j.jaccao.2024.07.017. eCollection 2024 Oct. JACC CardioOncol. 2024. PMID: 39479317 Free PMC article. Review.
-
Examining factors associated with experiencing cardiac arrhythmias in Black and White breast cancer survivors who received anthracyclines or trastuzumab.Breast Cancer Res Treat. 2025 Jun;211(2):537-544. doi: 10.1007/s10549-025-07671-0. Epub 2025 Mar 27. Breast Cancer Res Treat. 2025. PMID: 40146434 Free PMC article.
-
Testing home-based exercise strategies in underserved minority cancer patients undergoing chemotherapy (THRIVE) trial: a study protocol.Front Oncol. 2024 Sep 16;14:1427046. doi: 10.3389/fonc.2024.1427046. eCollection 2024. Front Oncol. 2024. PMID: 39351353 Free PMC article.
-
Biomarkers of Trastuzumab-Induced Cardiac Toxicity in HER2- Positive Breast Cancer Patient Population.Cancers (Basel). 2022 Jul 10;14(14):3353. doi: 10.3390/cancers14143353. Cancers (Basel). 2022. PMID: 35884413 Free PMC article.
-
Lifestyle factors in Black female breast cancer survivors-Descriptive results from an online pilot study.Front Public Health. 2023 Mar 14;11:1072741. doi: 10.3389/fpubh.2023.1072741. eCollection 2023. Front Public Health. 2023. PMID: 36998282 Free PMC article.
References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66(1):7–30. - PubMed
-
- Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344(11):783–792. - PubMed
-
- Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235(4785):177–182. - PubMed
-
- Perez EA, Romond EH, Suman VJ, Jeong JH, Davidson NE, Geyer CE, Jr, Martino S, Mamounas EP, Kaufman PA, Wolmark N. Four-Year Follow-Up of Trastuzumab Plus Adjuvant Chemotherapy for Operable Human Epidermal Growth Factor Receptor 2-Positive Breast Cancer: Joint Analysis of Data From NCCTG N9831 and NSABP B-31. J Clin Oncol. 2011;29(25):3366–73. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous