Comparison of the efficacy of a commercial inactivated influenza A/H1N1/pdm09 virus (pH1N1) vaccine and two experimental M2e-based vaccines against pH1N1 challenge in the growing pig model
- PMID: 29381710
- PMCID: PMC5790244
- DOI: 10.1371/journal.pone.0191739
Comparison of the efficacy of a commercial inactivated influenza A/H1N1/pdm09 virus (pH1N1) vaccine and two experimental M2e-based vaccines against pH1N1 challenge in the growing pig model
Abstract
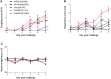
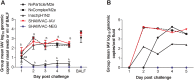
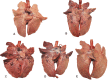
Swine influenza A viruses (IAV-S) found in North American pigs are diverse and the lack of cross-protection among heterologous strains is a concern. The objective of this study was to compare a commercial inactivated A/H1N1/pdm09 (pH1N1) vaccine and two novel subunit vaccines, using IAV M2 ectodomain (M2e) epitopes as antigens, in a growing pig model. Thirty-nine 2-week-old IAV negative pigs were randomly assigned to five groups and rooms. At 3 weeks of age and again at 5 weeks of age, pigs were vaccinated intranasally with an experimental subunit particle vaccine (NvParticle/M2e) or a subunit complex-based vaccine (NvComplex/M2e) or intramuscularly with a commercial inactivated vaccine (Inact/pH1N1). At 7 weeks of age, the pigs were challenged with pH1N1 virus or sham-inoculated. Necropsy was conducted 5 days post pH1N1 challenge (dpc). At the time of challenge one of the Inact/pH1N1 pigs had seroconverted based on IAV nucleoprotein-based ELISA, Inact/pH1N1 pigs had significantly higher pdm09H1N1 hemagglutination inhibition (HI) titers compared to all other groups, and M2e-specific IgG responses were detected in the NvParticle/M2e and the NvComplex/M2e pigs with significantly higher group means in the NvComplex/M2e group compared to SHAMVAC-NEG pigs. After challenge, nasal IAV RNA shedding was significantly reduced in Inact/pH1N1 pigs compared to all other pH1N1 infected groups and this group also had reduced IAV RNA in oral fluids. The macroscopic lung lesions were characterized by mild-to-severe, multifocal-to-diffuse, cranioventral dark purple consolidated areas typical of IAV infection and were similar for NvParticle/M2e, NvComplex/M2e and SHAMVAC-IAV pigs. Lesions were significantly less severe in the SHAMVAC-NEG and the Inact/pH1N1pigs. Under the conditions of this study, a commercial Inact/pH1N1 specific vaccine effectively protected pigs against homologous challenge as evidenced by reduced clinical signs, virus shedding in nasal secretions and oral fluids and reduced macroscopic and microscopic lesions whereas intranasal vaccination with experimental M2e epitope-based subunit vaccines did not. The results further highlight the importance using IAV-S type specific vaccines in pigs.
Conflict of interest statement
Figures

Similar articles
-
A prime-boost concept using a T-cell epitope-driven DNA vaccine followed by a whole virus vaccine effectively protected pigs in the pandemic H1N1 pig challenge model.Vaccine. 2019 Jul 18;37(31):4302-4309. doi: 10.1016/j.vaccine.2019.06.044. Epub 2019 Jun 24. Vaccine. 2019. PMID: 31248687
-
An experimental universal swine influenza a virus (IAV) vaccine candidate based on the M2 ectodomain (M2e) peptide does not provide protection against H1N1 IAV challenge in pigs.Vaccine. 2024 Jan 12;42(2):220-228. doi: 10.1016/j.vaccine.2023.12.012. Epub 2023 Dec 11. Vaccine. 2024. PMID: 38087714
-
Poly I:C adjuvanted inactivated swine influenza vaccine induces heterologous protective immunity in pigs.Vaccine. 2015 Jan 15;33(4):542-8. doi: 10.1016/j.vaccine.2014.11.034. Epub 2014 Nov 28. Vaccine. 2015. PMID: 25437101 Free PMC article.
-
Influenza A Virus in Swine: Epidemiology, Challenges and Vaccination Strategies.Front Vet Sci. 2020 Sep 22;7:647. doi: 10.3389/fvets.2020.00647. eCollection 2020. Front Vet Sci. 2020. PMID: 33195504 Free PMC article. Review.
-
Does Vaccine-Induced Maternally-Derived Immunity Protect Swine Offspring against Influenza a Viruses? A Systematic Review and Meta-Analysis of Challenge Trials from 1990 to May 2021.Animals (Basel). 2023 Oct 3;13(19):3085. doi: 10.3390/ani13193085. Animals (Basel). 2023. PMID: 37835692 Free PMC article. Review.
Cited by
-
An effective vaccine against influenza A virus based on the matrix protein 2 (M2).Vet Microbiol. 2024 Nov;298:110245. doi: 10.1016/j.vetmic.2024.110245. Epub 2024 Sep 14. Vet Microbiol. 2024. PMID: 39293153
References
-
- Gaymard A, Le BN, Frobert E, Lina B, Escuret V. Functional balance between neuraminidase and haemagglutinin in influenza viruses. Clin Microbiol Infect. 2016;22: 975–983. doi: 10.1016/j.cmi.2016.07.007 - DOI - PubMed
-
- Janke BH. Clinicopathological features of Swine influenza. Curr Top Microbiol Immunol. 2013;370: 69–83. doi: 10.1007/82_2013_308 - DOI - PubMed
-
- Forgie SE, Keenliside J, Wilkinson C, Webby R, Lu P, Sorensen O, et al. Swine outbreak of pandemic influenza A virus on a Canadian research farm supports human-to-swine transmission. Clin Infect Dis. 2011;52: 10–18. doi: 10.1093/cid/ciq030 - DOI - PMC - PubMed
-
- Neumann G, Noda T, Kawaoka Y. Emergence and pandemic potential of swine-origin H1N1 influenza virus. Nature. 2009;459: 931–939. doi: 10.1038/nature08157 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

