Efficacy of Epratuzumab, an Anti-CD22 Monoclonal IgG Antibody, in Systemic Lupus Erythematosus Patients With Associated Sjögren's Syndrome: Post Hoc Analyses From the EMBODY Trials
- PMID: 29381843
- PMCID: PMC5947119
- DOI: 10.1002/art.40425
Efficacy of Epratuzumab, an Anti-CD22 Monoclonal IgG Antibody, in Systemic Lupus Erythematosus Patients With Associated Sjögren's Syndrome: Post Hoc Analyses From the EMBODY Trials
Abstract
Objective: EMBODY 1 (ClinicalTrials.gov identifier: NCT01262365) and EMBODY 2 (ClinicalTrials.gov identifier: NCT01261793) investigated the efficacy and safety of epratuzumab, a CD22-targeted humanized monoclonal IgG antibody, in patients with systemic lupus erythematosus (SLE). The studies showed no significant difference from placebo in primary or secondary clinical outcome measures but did demonstrate B cell-specific immunologic activity. The aim of this post hoc analysis was to determine whether epratuzumab had a different clinical efficacy profile in SLE patients with versus those without an associated diagnosis of Sjögren's syndrome (SS).
Methods: The efficacy and safety of epratuzumab were compared between 2 patient subpopulations randomized in EMBODY 1 and 2: SLE patients with and those without a diagnosis of associated SS. British Isles Lupus Assessment Group (BILAG) total score, BILAG-based Combined Lupus Assessment (BICLA) clinical response to treatment, biologic markers (including B cells, IgG, IgM, and IgA), and safety were assessed.
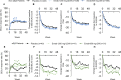
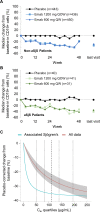
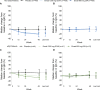
Results: A total of 1,584 patients were randomized in the EMBODY 1 and EMBODY 2 trials; 113 patients were anti-SSA positive and had a diagnosis of associated SS, and 1,375 patients (86.8%) had no diagnosis of associated SS (918 patients were randomized to receive epratuzumab and 457 to receive placebo). For patients with associated SS, but not those without associated SS, a higher proportion of patients receiving epratuzumab achieved a BICLA response and a reduction from baseline in BILAG total score. B cell reduction was faster in patients with associated SS. The sensitivity of B cells to epratuzumab as measured by the mean concentration producing 50% of the maximum B cell count depletion was lower for patients with associated SS (9.5 μg/ml) versus the total EMBODY population (87.1 μg/ml). No difference in the frequency of adverse events in those receiving placebo was reported.
Conclusion: Patients with SLE and associated SS treated with epratuzumab showed improvement in SLE disease activity, which was associated with bioactivity, such as decreases in B cell number and IgM level.
© 2018 The Authors. Arthritis & Rheumatology published by Wiley Periodicals, Inc. on behalf of American College of Rheumatology.
Figures

Comment in
-
Editorial: Epratuzumab: Reveille or Requiem? Teachable Moments for Lupus and Sjögren's Syndrome Clinical Trials.Arthritis Rheumatol. 2018 May;70(5):633-636. doi: 10.1002/art.40427. Epub 2018 Apr 16. Arthritis Rheumatol. 2018. PMID: 29381841 No abstract available.
Similar articles
-
Epratuzumab for the treatment of systemic lupus erythematosus.Expert Rev Clin Immunol. 2018 Apr;14(4):245-258. doi: 10.1080/1744666X.2018.1450141. Epub 2018 Mar 20. Expert Rev Clin Immunol. 2018. PMID: 29542345 Review.
-
Efficacy and Safety of Epratuzumab in Moderately to Severely Active Systemic Lupus Erythematosus: Results From Two Phase III Randomized, Double-Blind, Placebo-Controlled Trials.Arthritis Rheumatol. 2017 Feb;69(2):362-375. doi: 10.1002/art.39856. Arthritis Rheumatol. 2017. PMID: 27598855 Free PMC article. Clinical Trial.
-
Epratuzumab (humanised anti-CD22 antibody) in autoimmune diseases.Expert Opin Biol Ther. 2006 Sep;6(9):943-9. doi: 10.1517/14712598.6.9.943. Expert Opin Biol Ther. 2006. PMID: 16918261 Review.
-
Evaluation of epratuzumab as a biologic therapy in systemic lupus erythematosus.Immunotherapy. 2014;6(11):1165-75. doi: 10.2217/imt.14.80. Immunotherapy. 2014. PMID: 25496332 Review.
-
Efficacy and safety of epratuzumab in patients with moderate/severe active systemic lupus erythematosus: results from EMBLEM, a phase IIb, randomised, double-blind, placebo-controlled, multicentre study.Ann Rheum Dis. 2014 Jan;73(1):183-90. doi: 10.1136/annrheumdis-2012-202760. Epub 2013 Jan 12. Ann Rheum Dis. 2014. PMID: 23313811 Free PMC article. Clinical Trial.
Cited by
-
The full range of ophthalmological clinical manifestations in systemic lupus erythematosus.Front Ophthalmol (Lausanne). 2023 Jan 26;2:1055766. doi: 10.3389/fopht.2022.1055766. eCollection 2022. Front Ophthalmol (Lausanne). 2023. PMID: 38983519 Free PMC article.
-
Conformationally Constrained Sialyl Analogues as New Potential Binders of h-CD22.Chembiochem. 2022 May 18;23(10):e202200076. doi: 10.1002/cbic.202200076. Epub 2022 Mar 30. Chembiochem. 2022. PMID: 35313057 Free PMC article.
-
Distinct patterns of disease activity over time in patients with active SLE revealed using latent class trajectory models.Arthritis Res Ther. 2021 Jul 29;23(1):203. doi: 10.1186/s13075-021-02584-x. Arthritis Res Ther. 2021. PMID: 34321096 Free PMC article.
-
Peripheral B Cell Subsets in Autoimmune Diseases: Clinical Implications and Effects of B Cell-Targeted Therapies.J Immunol Res. 2020 Mar 23;2020:9518137. doi: 10.1155/2020/9518137. eCollection 2020. J Immunol Res. 2020. PMID: 32280720 Free PMC article. Review.
-
CD22: A Regulator of Innate and Adaptive B Cell Responses and Autoimmunity.Front Immunol. 2018 Sep 28;9:2235. doi: 10.3389/fimmu.2018.02235. eCollection 2018. Front Immunol. 2018. PMID: 30323814 Free PMC article. Review.
References
-
- Fox RI. Sjogren's syndrome. Lancet 2005;366:321–31. - PubMed
-
- Youinou P, Devauchelle‐Pensec V, Pers JO. Significance of B cells and B cell clonality in Sjögren's syndrome. Arthritis Rheum 2010;62:2605–10. - PubMed
-
- Salomonsson S, Jonsson MV, Skarstein K, Brokstad KA, Hjelmström P, Wahren‐Herlenius M, et al. Cellular basis of ectopic germinal center formation and autoantibody production in the target organ of patients with Sjögren's syndrome. Arthritis Rheum 2003;48:3187–201. - PubMed
-
- Risselada AP, Looije MF, Kruize AA, Bijlsma JW, van Roon JA. The role of ectopic germinal centers in the immunopathology of primary Sjögren's syndrome: a systematic review. Semin Arthritis Rheum 2013;42:368–76. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

