Tau Positron Emission Tomography Imaging in Degenerative Parkinsonisms
- PMID: 29381890
- PMCID: PMC5790630
- DOI: 10.14802/jmd.17071
Tau Positron Emission Tomography Imaging in Degenerative Parkinsonisms
Abstract
In recent years, several radiotracers that selectively bind to pathological tau proteins have been developed. Evidence is emerging that binding patterns of in vivo tau positron emission tomography (PET) studies in Alzheimer's disease (AD) patients closely resemble the distribution patterns of known neurofibrillary tangle pathology, with the extent of tracer binding reflecting the clinical and pathological progression of AD. In Lewy body diseases (LBD), tau PET imaging has clearly revealed cortical tau burden with a distribution pattern distinct from AD and increased cortical binding within the LBD spectrum. In progressive supranuclear palsy, the globus pallidus and midbrain have shown increased binding most prominently. Tau PET patterns in patients with corticobasal syndrome are characterized by asymmetrical uptake in the motor cortex and underlying white matter, as well as in the basal ganglia. Even in the patients with multiple system atrophy, which is basically a synucleinopathy, 18F-flortaucipir, a widely used tau PET tracer, also binds to the atrophic posterior putamen, possibly due to off-target binding. These distinct patterns of tau-selective radiotracer binding in the various degenerative parkinsonisms suggest its utility as a potential imaging biomarker for the differential diagnosis of parkinsonisms.
Keywords: Tau; parkinsonism; positron emission tomography.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures
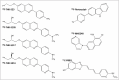

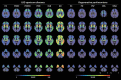
Similar articles
-
Imaging pathological tau in atypical parkinsonisms: A review.Clin Park Relat Disord. 2022 Jul 16;7:100155. doi: 10.1016/j.prdoa.2022.100155. eCollection 2022. Clin Park Relat Disord. 2022. PMID: 35880206 Free PMC article. Review.
-
Evaluation of [18F]PI-2620, a second-generation selective tau tracer, for assessing four-repeat tauopathies.Brain Commun. 2021 Aug 24;3(4):fcab190. doi: 10.1093/braincomms/fcab190. eCollection 2021. Brain Commun. 2021. PMID: 34632382 Free PMC article.
-
Subcortical 18 F-AV-1451 binding patterns in progressive supranuclear palsy.Mov Disord. 2017 Jan;32(1):134-140. doi: 10.1002/mds.26844. Epub 2016 Nov 3. Mov Disord. 2017. PMID: 27813160
-
Tau Imaging in Parkinsonism: What Have We Learned So Far?Mov Disord Clin Pract. 2018 Mar-Apr;5(2):118-130. doi: 10.1002/mdc3.12584. Epub 2017 Dec 29. Mov Disord Clin Pract. 2018. PMID: 30035155 Free PMC article.
-
Tau-PET imaging in Parkinson's disease: a systematic review and meta-analysis.Front Neurol. 2023 Apr 27;14:1145939. doi: 10.3389/fneur.2023.1145939. eCollection 2023. Front Neurol. 2023. PMID: 37181568 Free PMC article.
Cited by
-
What is New in Nuclear Medicine Imaging for Dementia.Noro Psikiyatr Ars. 2022 Dec 16;59(Suppl 1):S17-S23. doi: 10.29399/npa.28155. eCollection 2022. Noro Psikiyatr Ars. 2022. PMID: 36578980 Free PMC article. Review.
-
Integrated 18F-T807 Tau PET, Structural MRI, and Plasma Tau in Tauopathy Neurodegenerative Disorders.Front Aging Neurosci. 2021 Mar 29;13:646440. doi: 10.3389/fnagi.2021.646440. eCollection 2021. Front Aging Neurosci. 2021. PMID: 33854426 Free PMC article.
-
Tau Positron-Emission Tomography in Former National Football League Players.N Engl J Med. 2019 May 2;380(18):1716-1725. doi: 10.1056/NEJMoa1900757. Epub 2019 Apr 10. N Engl J Med. 2019. PMID: 30969506 Free PMC article.
-
Imaging pathological tau in atypical parkinsonisms: A review.Clin Park Relat Disord. 2022 Jul 16;7:100155. doi: 10.1016/j.prdoa.2022.100155. eCollection 2022. Clin Park Relat Disord. 2022. PMID: 35880206 Free PMC article. Review.
References
-
- Hatashita S, Yamasaki H, Suzuki Y, Tanaka K, Wakebe D, Hayakawa H. [18F]Flutemetamol amyloid-beta PET imaging compared with [11C]PIB across the spectrum of Alzheimer’s disease. Eur J Nucl Med Mol Imaging. 2014;41:290–300. - PubMed
-
- Rowe CC, Pejoska S, Mulligan RS, Jones G, Chan JG, Svensson S, et al. Head-to-head comparison of 11C-PiB and 18F-AZD4694 (NAV4694) for β-amyloid imaging in aging and dementia. J Nucl Med. 2013;54:880–886. - PubMed
-
- Villemagne VL, Mulligan RS, Pejoska S, Ong K, Jones G, O’Keefe G, et al. Comparison of 11C-PiB and 18F-florbetaben for Aβ imaging in ageing and Alzheimer’s disease. Eur J Nucl Med Mol Imaging. 2012;39:983–989. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

