Structure of tick-borne encephalitis virus and its neutralization by a monoclonal antibody
- PMID: 29382836
- PMCID: PMC5789857
- DOI: 10.1038/s41467-018-02882-0
Structure of tick-borne encephalitis virus and its neutralization by a monoclonal antibody
Abstract
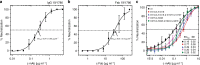
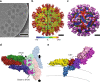
Tick-borne encephalitis virus (TBEV) causes 13,000 cases of human meningitis and encephalitis annually. However, the structure of the TBEV virion and its interactions with antibodies are unknown. Here, we present cryo-EM structures of the native TBEV virion and its complex with Fab fragments of neutralizing antibody 19/1786. Flavivirus genome delivery depends on membrane fusion that is triggered at low pH. The virion structure indicates that the repulsive interactions of histidine side chains, which become protonated at low pH, may contribute to the disruption of heterotetramers of the TBEV envelope and membrane proteins and induce detachment of the envelope protein ectodomains from the virus membrane. The Fab fragments bind to 120 out of the 180 envelope glycoproteins of the TBEV virion. Unlike most of the previously studied flavivirus-neutralizing antibodies, the Fab fragments do not lock the E-proteins in the native-like arrangement, but interfere with the process of virus-induced membrane fusion.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



Similar articles
-
Molecular Basis of a Protective/Neutralizing Monoclonal Antibody Targeting Envelope Proteins of both Tick-Borne Encephalitis Virus and Louping Ill Virus.J Virol. 2019 Apr 3;93(8):e02132-18. doi: 10.1128/JVI.02132-18. Print 2019 Apr 15. J Virol. 2019. PMID: 30760569 Free PMC article.
-
Molecular Organisation of Tick-Borne Encephalitis Virus.Viruses. 2022 Apr 11;14(4):792. doi: 10.3390/v14040792. Viruses. 2022. PMID: 35458522 Free PMC article.
-
Tick-Borne Encephalitis Virus: A Structural View.Viruses. 2018 Jun 28;10(7):350. doi: 10.3390/v10070350. Viruses. 2018. PMID: 29958443 Free PMC article. Review.
-
Antiviral activity of porphyrins and porphyrin-like compounds against tick-borne encephalitis virus: Blockage of the viral entry/fusion machinery by photosensitization-mediated destruction of the viral envelope.Antiviral Res. 2024 Jan;221:105767. doi: 10.1016/j.antiviral.2023.105767. Epub 2023 Nov 30. Antiviral Res. 2024. PMID: 38040199
-
[Molecular genetic characteristics of tick-borne encephalitis virus].Vopr Virusol. 2007 Sep-Oct;52(5):10-6. Vopr Virusol. 2007. PMID: 18041218 Review. Russian.
Cited by
-
Mechanism of differential Zika and dengue virus neutralization by a public antibody lineage targeting the DIII lateral ridge.J Exp Med. 2020 Feb 3;217(2):e20191792. doi: 10.1084/jem.20191792. J Exp Med. 2020. PMID: 31757867 Free PMC article.
-
Horses as Sentinels for the Circulation of Flaviviruses in Eastern-Central Germany.Viruses. 2023 Apr 30;15(5):1108. doi: 10.3390/v15051108. Viruses. 2023. PMID: 37243194 Free PMC article.
-
Exploring Tick-Borne Encephalitis Virus-Host Cell Interactions Using Electron Tomography: Methodologies and Protocols.Methods Mol Biol. 2025;2936:137-150. doi: 10.1007/978-1-0716-4587-1_15. Methods Mol Biol. 2025. PMID: 40512317
-
Tick-Borne Encephalitis (TBE): From Tick to Pathology.J Clin Med. 2023 Oct 30;12(21):6859. doi: 10.3390/jcm12216859. J Clin Med. 2023. PMID: 37959323 Free PMC article. Review.
-
Comparison of Extraction Methods for the Detection of Tick-Borne Encephalitis Virus RNA in Goat Raw Milk and Cream Cheese.Food Environ Virol. 2023 Mar;15(1):32-42. doi: 10.1007/s12560-022-09535-y. Epub 2022 Sep 20. Food Environ Virol. 2023. PMID: 36127560 Free PMC article.
References
-
- Suss J. Tick-borne encephalitis in Europe and beyond—the epidemiological situation as of 2007. Eur. Surveill. 2008;13:2–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

