Chronic inhalation of e-cigarette vapor containing nicotine disrupts airway barrier function and induces systemic inflammation and multiorgan fibrosis in mice
- PMID: 29384700
- PMCID: PMC6032308
- DOI: 10.1152/ajpregu.00270.2017
Chronic inhalation of e-cigarette vapor containing nicotine disrupts airway barrier function and induces systemic inflammation and multiorgan fibrosis in mice
Erratum in
-
Corrigendum for Alexander et al., volume 314, 2018, p. R834-R847.Am J Physiol Regul Integr Comp Physiol. 2022 Oct 1;323(4):R483. doi: 10.1152/ajpregu.00270.2017_cor. Am J Physiol Regul Integr Comp Physiol. 2022. PMID: 36169414 Free PMC article. No abstract available.
Abstract
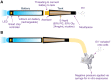
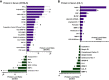
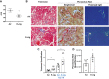
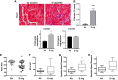
Electronic (e)-cigarettes theoretically may be safer than conventional tobacco. However, our prior studies demonstrated direct adverse effects of e-cigarette vapor (EV) on airway cells, including decreased viability and function. We hypothesize that repetitive, chronic inhalation of EV will diminish airway barrier function, leading to inflammatory protein release into circulation, creating a systemic inflammatory state, ultimately leading to distant organ injury and dysfunction. C57BL/6 and CD-1 mice underwent nose only EV exposure daily for 3-6 mo, followed by cardiorenal physiological testing. Primary human bronchial epithelial cells were grown at an air-liquid interface and exposed to EV for 15 min daily for 3-5 days before functional testing. Daily inhalation of EV increased circulating proinflammatory and profibrotic proteins in both C57BL/6 and CD-1 mice: the greatest increases observed were in angiopoietin-1 (31-fold) and EGF (25-fold). Proinflammatory responses were recapitulated by daily EV exposures in vitro of human airway epithelium, with EV epithelium secreting higher IL-8 in response to infection (227 vs. 37 pg/ml, respectively; P < 0.05). Chronic EV inhalation in vivo reduced renal filtration by 20% ( P = 0.017). Fibrosis, assessed by Masson's trichrome and Picrosirius red staining, was increased in EV kidneys (1.86-fold, C57BL/6; 3.2-fold, CD-1; P < 0.05), heart (2.75-fold, C57BL/6 mice; P < 0.05), and liver (1.77-fold in CD-1; P < 0.0001). Gene expression changes demonstrated profibrotic pathway activation. EV inhalation altered cardiovascular function, with decreased heart rate ( P < 0.01), and elevated blood pressure ( P = 0.016). These data demonstrate that chronic inhalation of EV may lead to increased inflammation, organ damage, and cardiorenal and hepatic disease.
Keywords: cardiorenal dysfunction; e-cigarette; electronic cigarette; fibrosis; nicotine; systemic inflammation.
Figures




References
-
- Alasmari F, Crotty Alexander LE, Nelson JA, Schiefer IT, Breen E, Drummond CA, Sari Y. Effects of chronic inhalation of electronic cigarettes containing nicotine on glial glutamate transporters and α-7 nicotinic acetylcholine receptor in female CD-1 mice. Prog Neuropsychopharmacol Biol Psychiatry 77: 1–8, 2017. doi:10.1016/j.pnpbp.2017.03.017. - DOI - PMC - PubMed
-
- Balloy V, Varet H, Dillies MA, Proux C, Jagla B, Coppée JY, Tabary O, Corvol H, Chignard M, Guillot L. Normal and cystic fibrosis human bronchial epithelial cells infected with Pseudomonas aeruginosa exhibit distinct gene activation patterns. PLoS One 10: e0140979, 2015. doi:10.1371/journal.pone.0140979. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

