Cell Penetrating Peptides as Molecular Carriers for Anti-Cancer Agents
- PMID: 29385037
- PMCID: PMC6017757
- DOI: 10.3390/molecules23020295
Cell Penetrating Peptides as Molecular Carriers for Anti-Cancer Agents
Abstract
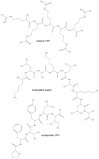
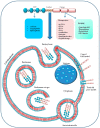
Cell membranes with their selective permeability play important functions in the tight control of molecular exchanges between the cytosol and the extracellular environment as the intracellular membranes do within the internal compartments. For this reason the plasma membranes often represent a challenging obstacle to the intracellular delivery of many anti-cancer molecules. The active transport of drugs through such barrier often requires specific carriers able to cross the lipid bilayer. Cell penetrating peptides (CPPs) are generally 5-30 amino acids long which, for their ability to cross cell membranes, are widely used to deliver proteins, plasmid DNA, RNA, oligonucleotides, liposomes and anti-cancer drugs inside the cells. In this review, we describe the several types of CPPs, the chemical modifications to improve their cellular uptake, the different mechanisms to cross cell membranes and their biological properties upon conjugation with specific molecules. Special emphasis has been given to those with promising application in cancer therapy.
Keywords: ">d-amino acids; cell penetrating peptides (CPPs); cellular uptake; chemical modifications; chemotherapeutic drugs; gene delivery; peptide cyclization; peptides; transfection.
Conflict of interest statement
The authors have declared no conflicts of interest.
Figures
Similar articles
-
Membrane Crossing and Membranotropic Activity of Cell-Penetrating Peptides: Dangerous Liaisons?Acc Chem Res. 2017 Dec 19;50(12):2968-2975. doi: 10.1021/acs.accounts.7b00455. Epub 2017 Nov 27. Acc Chem Res. 2017. PMID: 29172443
-
The Current Role of Cell-Penetrating Peptides in Cancer Therapy.Adv Exp Med Biol. 2017;1030:279-295. doi: 10.1007/978-3-319-66095-0_13. Adv Exp Med Biol. 2017. PMID: 29081059 Review.
-
Peptide vectors for the nonviral delivery of nucleic acids.Acc Chem Res. 2012 Jul 17;45(7):1048-56. doi: 10.1021/ar2002304. Epub 2012 Mar 28. Acc Chem Res. 2012. PMID: 22455499
-
Recent developments in anticancer drug delivery using cell penetrating and tumor targeting peptides.J Control Release. 2017 Mar 28;250:62-76. doi: 10.1016/j.jconrel.2017.02.006. Epub 2017 Feb 4. J Control Release. 2017. PMID: 28167286 Review.
-
Nanoparticles Modified with Cell-Penetrating Peptides: Conjugation Mechanisms, Physicochemical Properties, and Application in Cancer Diagnosis and Therapy.Int J Mol Sci. 2020 Apr 6;21(7):2536. doi: 10.3390/ijms21072536. Int J Mol Sci. 2020. PMID: 32268473 Free PMC article. Review.
Cited by
-
Cell-Penetrating Peptides Derived from Animal Venoms and Toxins.Toxins (Basel). 2021 Feb 15;13(2):147. doi: 10.3390/toxins13020147. Toxins (Basel). 2021. PMID: 33671927 Free PMC article. Review.
-
How well does molecular simulation reproduce environment-specific conformations of the intrinsically disordered peptides PLP, TP2 and ONEG?Chem Sci. 2022 Jan 20;13(7):1957-1971. doi: 10.1039/d1sc03496k. eCollection 2022 Feb 16. Chem Sci. 2022. PMID: 35308859 Free PMC article.
-
ACPred: A Computational Tool for the Prediction and Analysis of Anticancer Peptides.Molecules. 2019 May 22;24(10):1973. doi: 10.3390/molecules24101973. Molecules. 2019. PMID: 31121946 Free PMC article.
-
Synthesis, Characterization and Evaluation of Peptide Nanostructures for Biomedical Applications.Molecules. 2021 Jul 29;26(15):4587. doi: 10.3390/molecules26154587. Molecules. 2021. PMID: 34361740 Free PMC article. Review.
-
Potential of Cell-Penetrating Peptide-Conjugated Antisense Oligonucleotides for the Treatment of SMA.Molecules. 2024 Jun 4;29(11):2658. doi: 10.3390/molecules29112658. Molecules. 2024. PMID: 38893532 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

