Contrasting impacts of competition on ecological and social trait evolution in songbirds
- PMID: 29385141
- PMCID: PMC5809094
- DOI: 10.1371/journal.pbio.2003563
Contrasting impacts of competition on ecological and social trait evolution in songbirds
Abstract
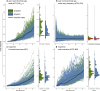
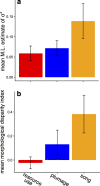
Competition between closely related species has long been viewed as a powerful selective force that drives trait diversification, thereby generating phenotypic diversity over macroevolutionary timescales. However, although the impact of interspecific competition has been documented in a handful of iconic insular radiations, most previous studies have focused on traits involved in resource use, and few have examined the role of competition across large, continental radiations. Thus, the extent to which broad-scale patterns of phenotypic diversity are shaped by competition remain largely unclear, particularly for social traits. Here, we estimate the effect of competition between interacting lineages by applying new phylogenetic models that account for such interactions to an exceptionally complete dataset of resource-use traits and social signaling traits for the entire radiation of tanagers (Aves, Thraupidae), the largest family of songbirds. We find that interspecific competition strongly influences the evolution of traits involved in resource use, with a weaker effect on plumage signals, and very little effect on song. Our results provide compelling evidence that interspecific exploitative competition contributes to ecological trait diversification among coexisting species, even in a large continental radiation. In comparison, signal traits mediating mate choice and social competition seem to diversify under different evolutionary models, including rapid diversification in the allopatric stage of speciation.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Darwin C. On the origin of the species by natural selection. Murray; 1859.
-
- Pfennig DW, Pfennig KS. Evolution’s Wedge: Competition and the Origins of Diversity. Los Angeles, CA: Univ of California Press; 2012.
-
- Schluter D. Ecological character displacement in adaptive radiation. Am Nat. 2000;156: S4–S16. doi: 10.1086/303412 - DOI
-
- Grant PR, Grant BR. Evolution of character displacement in Darwin’s finches. Science. 2006;313: 224–226. doi: 10.1126/science.1128374 - DOI - PubMed
-
- Schluter D, Grant PR. Determinants of Morphological Patterns in Communities of Darwin’s Finches. Am Nat. 1984;123: 175–196. doi: 10.1086/284196 - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

