Giardia secretome highlights secreted tenascins as a key component of pathogenesis
- PMID: 29385462
- PMCID: PMC5887430
- DOI: 10.1093/gigascience/giy003
Giardia secretome highlights secreted tenascins as a key component of pathogenesis
Abstract
Background: Giardia is a protozoan parasite of public health relevance that causes gastroenteritis in a wide range of hosts. Two genetically distinct lineages (assemblages A and B) are responsible for the human disease. Although it is clear that differences in virulence occur, the pathogenesis and virulence of Giardia remain poorly understood.
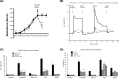
Results: The genome of Giardia is believed to contain open reading frames that could encode as many as 6000 proteins. By successfully applying quantitative proteomic analyses to the whole parasite and to the supernatants derived from parasite culture of assemblages A and B, we confirm expression of ∼1600 proteins from each assemblage, the vast majority of which are common to both lineages. To look for signature enrichment of secreted proteins, we considered the ratio of proteins in the supernatant compared with the pellet, which defined a small group of enriched proteins, putatively secreted at a steady state by cultured growing trophozoites of both assemblages. This secretome is enriched with proteins annotated to have N-terminal signal peptide. The most abundant secreted proteins include known virulence factors such as cathepsin B cysteine proteases and members of a Giardia superfamily of cysteine-rich proteins that comprise variant surface proteins, high-cysteine membrane proteins, and a new class of virulence factors, the Giardia tenascins. We demonstrate that physiological function of human enteric epithelial cells is disrupted by such soluble factors even in the absence of the trophozoites.
Conclusions: We are able to propose a straightforward model of Giardia pathogenesis incorporating key roles for the major Giardia-derived soluble mediators.
Figures

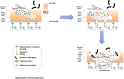
 ) renders the intestinal environment more favourable to trophozoite's growth. Once a new Giardia colony is established, trophozoites release extracellular nuclease (
) renders the intestinal environment more favourable to trophozoite's growth. Once a new Giardia colony is established, trophozoites release extracellular nuclease ( ), GCATB (
), GCATB ( ), and tenascin (
), and tenascin ( ). Extracellular nuclease may contribute to reducing the viscosity of the intestinal outer mucus layer, while GCATB may degrade mucins and disrupt intracellular junction. Finally, tenascins may maintain intestinal cells apart by attaching to the EGF receptors present at the surface of intestinal cells that could, over time, lead to the apoptosis of these isolated intestinal cells.
). Extracellular nuclease may contribute to reducing the viscosity of the intestinal outer mucus layer, while GCATB may degrade mucins and disrupt intracellular junction. Finally, tenascins may maintain intestinal cells apart by attaching to the EGF receptors present at the surface of intestinal cells that could, over time, lead to the apoptosis of these isolated intestinal cells.Similar articles
-
Giardia duodenalis Virulence - "To Be, or Not To Be".Curr Trop Med Rep. 2021;8(4):246-256. doi: 10.1007/s40475-021-00248-z. Epub 2021 Oct 21. Curr Trop Med Rep. 2021. PMID: 34697581 Free PMC article. Review.
-
Giardia duodenalis: Role of secreted molecules as virulent factors in the cytotoxic effect on epithelial cells.Adv Parasitol. 2019;106:129-169. doi: 10.1016/bs.apar.2019.07.003. Epub 2019 Aug 20. Adv Parasitol. 2019. PMID: 31630757 Review.
-
Induction of virulence factors in Giardia duodenalis independent of host attachment.Sci Rep. 2016 Feb 12;6:20765. doi: 10.1038/srep20765. Sci Rep. 2016. PMID: 26867958 Free PMC article.
-
Characterization of the Giardia intestinalis secretome during interaction with human intestinal epithelial cells: The impact on host cells.PLoS Negl Trop Dis. 2017 Dec 11;11(12):e0006120. doi: 10.1371/journal.pntd.0006120. eCollection 2017 Dec. PLoS Negl Trop Dis. 2017. PMID: 29228011 Free PMC article.
-
Genotyping and Descriptive Proteomics of a Potential Zoonotic Canine Strain of Giardia duodenalis, Infective to Mice.PLoS One. 2016 Oct 19;11(10):e0164946. doi: 10.1371/journal.pone.0164946. eCollection 2016. PLoS One. 2016. PMID: 27760188 Free PMC article.
Cited by
-
Insights into Peptidyl-Prolyl cis-trans Isomerases from Clinically Important Protozoans: From Structure to Potential Biotechnological Applications.Pathogens. 2024 Jul 31;13(8):644. doi: 10.3390/pathogens13080644. Pathogens. 2024. PMID: 39204244 Free PMC article. Review.
-
Giardia duodenalis Virulence - "To Be, or Not To Be".Curr Trop Med Rep. 2021;8(4):246-256. doi: 10.1007/s40475-021-00248-z. Epub 2021 Oct 21. Curr Trop Med Rep. 2021. PMID: 34697581 Free PMC article. Review.
-
Identification of Conserved Candidate Vaccine Antigens in the Surface Proteome of Giardia lamblia.Infect Immun. 2019 May 21;87(6):e00219-19. doi: 10.1128/IAI.00219-19. Print 2019 Jun. Infect Immun. 2019. PMID: 30962402 Free PMC article.
-
Global epidemiology of Giardia duodenalis infection in cancer patients: a systematic review and meta-analysis.Int Health. 2022 Jan 19;14(1):5-17. doi: 10.1093/inthealth/ihab026. Int Health. 2022. PMID: 34022771 Free PMC article.
-
The Cysteine Protease Giardipain-1 from Giardia duodenalis Contributes to a Disruption of Intestinal Homeostasis.Int J Mol Sci. 2022 Nov 7;23(21):13649. doi: 10.3390/ijms232113649. Int J Mol Sci. 2022. PMID: 36362435 Free PMC article.
References
-
- Nash TE, Herrington DA, Losonsky GA et al. . Experimental human infections with Giardia lamblia. J Infect Dis 1987;156(6):974–84. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

