Evaluating In Vitro-In Vivo Extrapolation of Toxicokinetics
- PMID: 29385628
- PMCID: PMC5920326
- DOI: 10.1093/toxsci/kfy020
Evaluating In Vitro-In Vivo Extrapolation of Toxicokinetics
Abstract
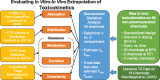
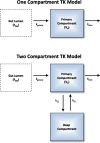
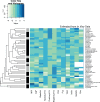
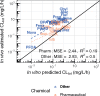
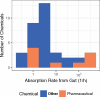
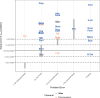
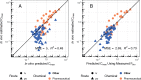
Prioritizing the risk posed by thousands of chemicals potentially present in the environment requires exposure, toxicity, and toxicokinetic (TK) data, which are often unavailable. Relatively high throughput, in vitro TK (HTTK) assays and in vitro-to-in vivo extrapolation (IVIVE) methods have been developed to predict TK, but most of the in vivo TK data available to benchmark these methods are from pharmaceuticals. Here we report on new, in vivo rat TK experiments for 26 non-pharmaceutical chemicals with environmental relevance. Both intravenous and oral dosing were used to calculate bioavailability. These chemicals, and an additional 19 chemicals (including some pharmaceuticals) from previously published in vivo rat studies, were systematically analyzed to estimate in vivo TK parameters (e.g., volume of distribution [Vd], elimination rate). For each of the chemicals, rat-specific HTTK data were available and key TK predictions were examined: oral bioavailability, clearance, Vd, and uncertainty. For the non-pharmaceutical chemicals, predictions for bioavailability were not effective. While no pharmaceutical was absorbed at less than 10%, the fraction bioavailable for non-pharmaceutical chemicals was as low as 0.3%. Total clearance was generally more under-estimated for nonpharmaceuticals and Vd methods calibrated to pharmaceuticals may not be appropriate for other chemicals. However, the steady-state, peak, and time-integrated plasma concentrations of nonpharmaceuticals were predicted with reasonable accuracy. The plasma concentration predictions improved when experimental measurements of bioavailability were incorporated. In summary, HTTK and IVIVE methods are adequately robust to be applied to high throughput in vitro toxicity screening data of environmentally relevant chemicals for prioritizing based on human health risks.
Figures




References
-
- Akaike H. (1974). A new look at the statistical model identification. IEEE Tran. Automat. Control 19, 716–723.http://dx.doi.org/10.1109/TAC.1974.1100705 - DOI
-
- Andersen M. E., Clewell H. J., Tan Y.-M., Butenhoff J. L., Olsen G. W. (2006). Pharmacokinetic modeling of saturable, renal resorption of perfluoroalkylacids in monkeys—probing the determinants of long plasma half-lives. Toxicology 227, 156–164. - PubMed
-
- Armitage J. M., Wania F., Arnot J. A. (2014). Application of mass blanace models and the chemical activity concept to facilitate the use of in vitro toxicity data for risk assessment. Environ. Sci. Technol. 48, 9770–9779.http://dx.doi.org/10.1021/es501955g - DOI - PubMed
-
- Bartlett M. S. (1953a). Approximate confidence intervals. II. More than one unknown parameter. Biometrika 40, 306–317.
-
- Bartlett M. S. (1953b). Approximate confidence intervals. Biometrika 40, 12–19.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

