Ablation of the Right Cardiac Vagus Nerve Reduces Acetylcholine Content without Changing the Inflammatory Response during Endotoxemia
- PMID: 29389905
- PMCID: PMC5855664
- DOI: 10.3390/ijms19020442
Ablation of the Right Cardiac Vagus Nerve Reduces Acetylcholine Content without Changing the Inflammatory Response during Endotoxemia
Abstract
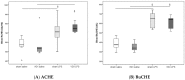
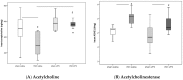
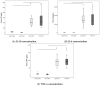
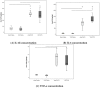
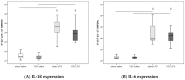
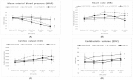
Acetylcholine is the main transmitter of the parasympathetic vagus nerve. According to the cholinergic anti-inflammatory pathway (CAP) concept, acetylcholine has been shown to be important for signal transmission within the immune system and also for a variety of other functions throughout the organism. The spleen is thought to play an important role in regulating the CAP. In contrast, the existence of a "non-neuronal cardiac cholinergic system" that influences cardiac innervation during inflammation has been hypothesized, with recent publications introducing the heart instead of the spleen as a possible interface between the immune and nervous systems. To prove this hypothesis, we investigated whether selectively disrupting vagal stimulation of the right ventricle plays an important role in rat CAP regulation during endotoxemia. We performed a selective resection of the right cardiac branch of the Nervus vagus (VGX) with a corresponding sham resection in vehicle-injected and endotoxemic rats. Rats were injected with lipopolysaccharide (LPS, 1 mg/kg body weight, intravenously) and observed for 4 h. Intraoperative blood gas analysis was performed, and hemodynamic parameters were assessed using a left ventricular pressure-volume catheter. Rat hearts and blood were collected, and the expression and concentration of proinflammatory cytokines using quantitative reverse transcription polymerase chain reaction and enzyme-linked immunosorbent assay were measured, respectively. Four hours after injection, LPS induced a marked deterioration in rat blood gas parameters such as pH value, potassium, base excess, glucose, and lactate. The mean arterial blood pressure and the end-diastolic volume had decreased significantly. Further, significant increases in blood cholinesterases and in proinflammatory (IL-1β, IL-6, TNF-α) cytokine concentration and gene expression were obtained. Right cardiac vagus nerve resection (VGX) led to a marked decrease in heart acetylcholine concentration and an increase in cardiac acetylcholinesterase activity. Without LPS, VGX changed rat hemodynamic parameters, including heart frequency, cardiac output, and end-diastolic volume. In contrast, VGX during endotoxemia did not significantly change the concentration and expression of proinflammatory cytokines in the heart. In conclusion we demonstrate that right cardiac vagal innervation regulates cardiac acetylcholine content but neither improves nor worsens systemic inflammation.
Keywords: endotoxemia; hemodynamic parameters; inflammation; right cardiac vagus nerve resection; sepsis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Loss of vagal tone aggravates systemic inflammation and cardiac impairment in endotoxemic rats.J Surg Res. 2014 May 15;188(2):480-8. doi: 10.1016/j.jss.2014.01.022. Epub 2014 Jan 21. J Surg Res. 2014. PMID: 24565505
-
Impaired vagus-mediated immunosuppression in microsomal prostaglandin E synthase-1 deficient mice.Prostaglandins Other Lipid Mediat. 2015 Sep;121(Pt B):155-62. doi: 10.1016/j.prostaglandins.2015.05.006. Epub 2015 May 19. Prostaglandins Other Lipid Mediat. 2015. PMID: 26001880
-
The role of spleen in vagus nerve stimulation for treatment against septic shock in rats.Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2011 May;23(5):263-6. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2011. PMID: 21805738
-
The cholinergic anti-inflammatory pathway: a critical review.Auton Neurosci. 2014 May;182:65-9. doi: 10.1016/j.autneu.2013.12.007. Epub 2013 Dec 24. Auton Neurosci. 2014. PMID: 24411268 Review.
-
Anti-inflammatory properties of the vagus nerve: potential therapeutic implications of vagus nerve stimulation.J Physiol. 2016 Oct 15;594(20):5781-5790. doi: 10.1113/JP271539. Epub 2016 May 1. J Physiol. 2016. PMID: 27059884 Free PMC article. Review.
Cited by
-
Neuronal Mitochondria Modulation of LPS-Induced Neuroinflammation.J Neurosci. 2020 Feb 19;40(8):1756-1765. doi: 10.1523/JNEUROSCI.2324-19.2020. Epub 2020 Jan 14. J Neurosci. 2020. PMID: 31937559 Free PMC article.
-
Olanzapine Modulate Lipid Metabolism and Adipose Tissue Accumulation via Hepatic Muscarinic M3 Receptor-Mediated Alk-Related Signaling.Biomedicines. 2024 Jun 25;12(7):1403. doi: 10.3390/biomedicines12071403. Biomedicines. 2024. PMID: 39061977 Free PMC article.
-
Proinflammatory Extracellular Vesicle-Mediated Signaling Contributes to the Induction of Neuroinflammation in Animal Models of Endotoxemia and Peripheral Surgical Stress.Cell Mol Neurobiol. 2021 Aug;41(6):1325-1336. doi: 10.1007/s10571-020-00905-3. Epub 2020 Jun 18. Cell Mol Neurobiol. 2021. PMID: 32557202 Free PMC article.
-
Research progress in myocardial function and diseases related to muscarinic acetylcholine receptor (Review).Int J Mol Med. 2025 Jun;55(6):86. doi: 10.3892/ijmm.2025.5527. Epub 2025 Apr 4. Int J Mol Med. 2025. PMID: 40183403 Free PMC article. Review.
-
Effects of Vitamin D Deficiency on the Function of the Cardiac Autonomic Nervous System in Rats.Cardiovasc Ther. 2022 Mar 23;2022:4366948. doi: 10.1155/2022/4366948. eCollection 2022. Cardiovasc Ther. 2022. PMID: 35387268 Free PMC article.
References
-
- Engel C., Brunkhorst F.M., Bone H.G., Brunkhorst R., Gerlach H., Grond S., Gruendling M., Huhle G., Jaschinski U., John S., et al. Epidemiology of sepsis in Germany: Results from a national prospective multicenter study. Intensive Care Med. 2007;33:606–618. doi: 10.1007/s00134-006-0517-7. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

