An oxidized abasic lesion inhibits base excision repair leading to DNA strand breaks in a trinucleotide repeat tract
- PMID: 29389977
- PMCID: PMC5794147
- DOI: 10.1371/journal.pone.0192148
An oxidized abasic lesion inhibits base excision repair leading to DNA strand breaks in a trinucleotide repeat tract
Abstract
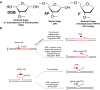
Oxidative DNA damage and base excision repair (BER) play important roles in modulating trinucleotide repeat (TNR) instability that is associated with human neurodegenerative diseases and cancer. We have reported that BER of base lesions can lead to TNR instability. However, it is unknown if modifications of the sugar in an abasic lesion modulate TNR instability. In this study, we characterized the effects of the oxidized sugar, 5'-(2-phosphoryl-1,4-dioxobutane)(DOB) in CAG repeat tracts on the activities of key BER enzymes, as well as on repeat instability. We found that DOB crosslinked with DNA polymerase β and inhibited its synthesis activity in CAG repeat tracts. Surprisingly, we found that DOB also formed crosslinks with DNA ligase I and inhibited its ligation activity, thereby reducing the efficiency of BER. This subsequently resulted in the accumulation of DNA strand breaks in a CAG repeat tract. Our study provides important new insights into the adverse effects of an oxidized abasic lesion on BER and suggests a potential alternate repair pathway through which an oxidized abasic lesion may modulate TNR instability.
Conflict of interest statement
Figures








References
-
- Paulson HL, Fischbeck KH. Trinucleotide repeats in neurogenetic disorders. Annu Rev Neurosci. 1996;19:79–107. doi: 10.1146/annurev.ne.19.030196.000455 - DOI - PubMed
-
- McMurray CT. Mechanisms of trinucleotide repeat instability during human development. Nat Rev Genet. 2010;11(11):786–99. doi: 10.1038/nrg2828 - DOI - PMC - PubMed
-
- Wells RD, Dere R, Hebert ML, Napierala M, Son LS. Advances in mechanisms of genetic instability related to hereditary neurological diseases. Nucleic Acids Res. 2005;33(12):3785–98. doi: 10.1093/nar/gki697 - DOI - PMC - PubMed
-
- Usdin K, House NC, Freudenreich CH. Repeat instability during DNA repair: Insights from model systems. Crit Rev Biochem Mol Biol. 2015;50(2):142–67. doi: 10.3109/10409238.2014.999192 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

