Enhancing Plasticity of the Central Nervous System: Drugs, Stem Cell Therapy, and Neuro-Implants
- PMID: 29391951
- PMCID: PMC5748136
- DOI: 10.1155/2017/2545736
Enhancing Plasticity of the Central Nervous System: Drugs, Stem Cell Therapy, and Neuro-Implants
Abstract
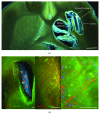
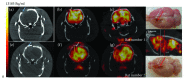
Stroke represents the first cause of adult acquired disability. Spontaneous recovery, dependent on endogenous neurogenesis, allows for limited recovery in 50% of patients who remain functionally dependent despite physiotherapy. Here, we propose a review of novel drug therapies with strong potential in the clinic. We will also discuss new avenues of stem cell therapy in patients with a cerebral lesion. A promising future for the development of efficient drugs to enhance functional recovery after stroke seems evident. These drugs will have to prove their efficacy also in severely affected patients. The efficacy of stem cell engraftment has been demonstrated but will have to prove its potential in restoring tissue function for the massive brain lesions that are most debilitating. New answers may lay in biomaterials, a steadily growing field. Biomaterials should ideally resemble lesioned brain structures in architecture and must be proven to increase functional reconnections within host tissue before clinical testing.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

