Glomerular endothelial cell maturation depends on ADAM10, a key regulator of Notch signaling
- PMID: 29397483
- PMCID: PMC5878725
- DOI: 10.1007/s10456-018-9599-4
Glomerular endothelial cell maturation depends on ADAM10, a key regulator of Notch signaling
Abstract
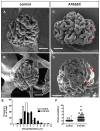
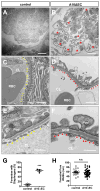
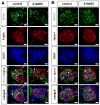
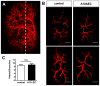
The principal function of glomeruli is to filter blood through a highly specialized filtration barrier consisting of a fenestrated endothelium, the glomerular basement membrane and podocyte foot processes. Previous studies have uncovered a crucial role of endothelial a disintegrin and metalloprotease 10 (ADAM10) and Notch signaling in the development of glomeruli, yet the resulting defects have not been further characterized nor understood in the context of kidney development. Here, we used several different experimental approaches to analyze the kidneys and glomeruli from mice lacking ADAM10 in endothelial cells (A10ΔEC mice). Scanning electron microscopy of glomerular casts demonstrated enlarged vascular diameter and increased intussusceptive events in A10ΔEC glomeruli compared to controls. Consistent with these findings, genes known to regulate vessel caliber (Apln, AplnR and Vegfr3) are significantly upregulated in A10ΔEC glomeruli. Moreover, transmission electron microscopy revealed the persistence of diaphragms in the fenestrae of A10ΔEC glomerular endothelial cells, which was corroborated by the elevated expression of the protein PLVAP/PV-1, an integral component of fenestral diaphragms. Analysis of gross renal vasculature by light sheet microscopy showed no major alteration of the branching pattern, indicating a localized importance of ADAM10 in the glomerular endothelium. Since intussusceptions and fenestrae with diaphragms are normally found in developing, but not mature glomeruli, our results provide the first evidence for a crucial role of endothelial ADAM10, a key regulator of Notch signaling, in promoting the development and maturation of the glomerular vasculature.
Keywords: A disintegrin and metalloprotease 10 (ADAM10); Diaphragms; Endothelial cells; Fenestra; Glomeruli; Notch.
Conflict of interest statement
Figures


Similar articles
-
Deletion of Adam10 in endothelial cells leads to defects in organ-specific vascular structures.Blood. 2011 Jul 28;118(4):1163-74. doi: 10.1182/blood-2011-04-348557. Epub 2011 Jun 7. Blood. 2011. PMID: 21652679 Free PMC article.
-
ADAM10 controls the differentiation of the coronary arterial endothelium.Angiogenesis. 2019 May;22(2):237-250. doi: 10.1007/s10456-018-9653-2. Epub 2018 Nov 16. Angiogenesis. 2019. PMID: 30446855 Free PMC article.
-
Endothelial deletion of ADAM10, a key regulator of Notch signaling, causes impaired decidualization and reduced fertility in female mice.Angiogenesis. 2020 Aug;23(3):443-458. doi: 10.1007/s10456-020-09723-z. Epub 2020 May 8. Angiogenesis. 2020. PMID: 32385775 Free PMC article.
-
Intriguing Roles for Endothelial ADAM10/Notch Signaling in the Development of Organ-Specific Vascular Beds.Physiol Rev. 2018 Oct 1;98(4):2025-2061. doi: 10.1152/physrev.00029.2017. Physiol Rev. 2018. PMID: 30067156 Free PMC article. Review.
-
Regulation of the trafficking and the function of the metalloprotease ADAM10 by tetraspanins.Biochem Soc Trans. 2017 Aug 15;45(4):937-44. doi: 10.1042/BST20160296. Epub 2017 Jul 7. Biochem Soc Trans. 2017. PMID: 28687716 Review.
Cited by
-
PLVAP is associated with glioma-associated malignant processes and immunosuppressive cell infiltration as a promising marker for prognosis.Heliyon. 2022 Aug 19;8(8):e10298. doi: 10.1016/j.heliyon.2022.e10298. eCollection 2022 Aug. Heliyon. 2022. PMID: 36033326 Free PMC article.
-
Phorbol esters induce PLVAP expression via VEGF and additional secreted molecules in MEK1-dependent and p38, JNK and PI3K/Akt-independent manner.J Cell Mol Med. 2019 Feb;23(2):920-933. doi: 10.1111/jcmm.13993. Epub 2018 Nov 5. J Cell Mol Med. 2019. PMID: 30394679 Free PMC article.
-
The role of PLVAP in endothelial cells.Cell Tissue Res. 2023 May;392(2):393-412. doi: 10.1007/s00441-023-03741-1. Epub 2023 Feb 13. Cell Tissue Res. 2023. PMID: 36781482 Free PMC article. Review.
-
Therapeutic contribution of melatonin to the treatment of septic cardiomyopathy: A novel mechanism linking Ripk3-modified mitochondrial performance and endoplasmic reticulum function.Redox Biol. 2019 Sep;26:101287. doi: 10.1016/j.redox.2019.101287. Epub 2019 Jul 27. Redox Biol. 2019. PMID: 31386965 Free PMC article.
-
The Life of a Kidney Podocyte.Acta Physiol (Oxf). 2025 Aug;241(8):e70081. doi: 10.1111/apha.70081. Acta Physiol (Oxf). 2025. PMID: 40698593 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

