A Virus-Like-Particle immunotherapy targeting Epitope-Specific anti-xCT expressed on cancer stem cell inhibits the progression of metastatic cancer in vivo
- PMID: 29399412
- PMCID: PMC5790338
- DOI: 10.1080/2162402X.2017.1408746
A Virus-Like-Particle immunotherapy targeting Epitope-Specific anti-xCT expressed on cancer stem cell inhibits the progression of metastatic cancer in vivo
Abstract
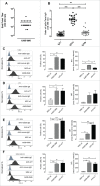
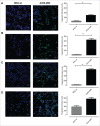
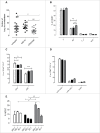
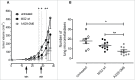
Aggressive forms of breast cancer, such as Her2+ and triple negative breast cancer (TNBC), are enriched in breast cancer stem cells (BCSC) and have limited therapeutic options. BCSC represent a key cellular reservoir for relapse, metastatic progression and therapeutic resistance. Their ability to resist common cytotoxic therapies relies on different mechanisms, including improved detoxification. The cystine-glutamate antiporter protein xCT (SLC7A11) regulates cystine intake, conversion to cysteine and subsequent glutathione synthesis, protecting cells against oxidative and chemical insults. Our previous work showed that xCT is highly expressed in tumorspheres derived from breast cancer cell lines and downregulation of xCT altered BCSC function in vitro and inhibited pulmonary metastases in vivo. We further strengthened these observations by developing a virus-like-particle (VLP; AX09-0M6) immunotherapy targeting the xCT protein. AX09-0M6 elicited a strong antibody response against xCT including high levels of IgG2a antibody. IgG isolated from AX09-0M6 treated mice bound to tumorspheres, inhibited xCT function as assessed by reactive oxygen species generation and decreased BCSC growth and self-renewal. To assess if AX09-0M6 impacts BCSC in vivo seeding, Her2+ TUBO-derived tumorspheres were injected into the tail vein of AX09-0M6 or control treated female BALB/c mice. AX09-0M6 significantly inhibited formation of pulmonary nodules. To evaluate its ability to impact metastases, AX09-0M6 was administered to mice with established subcutaneous 4T1 tumors. AX09-0M6 administration significantly hampered tumor growth and development of pulmonary metastases. These data show that a VLP-based immunization approach inhibits xCT activity, impacts BCSC biology and significantly reduces metastatic progression in preclinical models.
Keywords: Breast cancer; cancer stem cell; immunotherapy; virus like-particle; xCT.
Figures



References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
