Ribokinase from Leishmania donovani: purification, characterization and X-ray crystallographic analysis
- PMID: 29400319
- PMCID: PMC5947680
- DOI: 10.1107/S2053230X18000109
Ribokinase from Leishmania donovani: purification, characterization and X-ray crystallographic analysis
Abstract
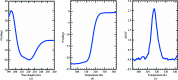
Leishmania is an auxotrophic protozoan parasite which acquires D-ribose by transporting it from the host cell and also by the hydrolysis of nucleosides. The enzyme ribokinase (RK) catalyzes the first step of ribose metabolism by phosphorylating D-ribose using ATP to produce D-ribose-5-phosphate. To understand its structure and function, the gene encoding RK from L. donovani was cloned, expressed and purified using affinity and size-exclusion chromatography. Circular-dichroism spectroscopy of the purified protein showed comparatively more α-helix in the secondary-structure content, and thermal unfolding revealed the Tm to be 317.2 K. Kinetic parameters were obtained by functional characterization of L. donovani RK, and the Km values for ribose and ATP were found to be 296 ± 36 and 116 ± 9.0 µM, respectively. Crystals obtained by the hanging-drop vapour-diffusion method diffracted to 1.95 Å resolution and belonged to the hexagonal space group P61, with unit-cell parameters a = b = 100.25, c = 126.77 Å. Analysis of the crystal content indicated the presence of two protomers in the asymmetric unit, with a Matthews coefficient (VM) of 2.45 Å3 Da-1 and 49.8% solvent content. Further study revealed that human counterpart of this protein could be used as a template to determine the first three-dimensional structure of the RK from trypanosomatid parasites.
Keywords: Leishmania donovani; crystallization; d-ribose metabolism; ribokinase; thermal stability.
Figures




Similar articles
-
Cloning, purification, crystallization and preliminary crystallographic analysis of a ribokinase from Staphylococcus aureus.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2009 Jun 1;65(Pt 6):574-6. doi: 10.1107/S1744309109014833. Epub 2009 May 22. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2009. PMID: 19478434 Free PMC article.
-
Crystallization and preliminary X-ray analysis of a ribokinase from Vibrio cholerae O395.Acta Crystallogr F Struct Biol Commun. 2014 Aug;70(Pt 8):1098-102. doi: 10.1107/S2053230X14014411. Epub 2014 Jul 23. Acta Crystallogr F Struct Biol Commun. 2014. PMID: 25084391 Free PMC article.
-
Unraveling structural insights of ribokinase from Leishmania donovani.Int J Biol Macromol. 2019 Sep 1;136:253-265. doi: 10.1016/j.ijbiomac.2019.06.001. Epub 2019 Jun 3. Int J Biol Macromol. 2019. PMID: 31170491
-
Expression, purification, crystallization and preliminary X-ray diffraction analysis of a ribokinase from the thermohalophile Halothermothrix orenii.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012 Feb 1;68(Pt 2):240-3. doi: 10.1107/S1744309111041091. Epub 2012 Jan 27. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012. PMID: 22298009 Free PMC article.
-
Crystallization and preliminary crystallographic analysis of the tetrameric form of phosphofructokinase-2 from Escherichia coli, a member of the ribokinase family.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2006 Sep 1;62(Pt 9):935-7. doi: 10.1107/S1744309106032246. Epub 2006 Aug 26. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2006. PMID: 16946484 Free PMC article.
Cited by
-
Biochemical and structural insights into 6-phosphogluconate dehydrogenase from Leishmania donovani.Appl Microbiol Biotechnol. 2021 Jul;105(13):5471-5489. doi: 10.1007/s00253-021-11434-4. Epub 2021 Jul 12. Appl Microbiol Biotechnol. 2021. PMID: 34250571
References
-
- Anderson, A. & Cooper, R. A. (1969). Biochim. Biophys. Acta, 177, 163–165. - PubMed
-
- Andersson, C. E. & Mowbray, S. L. (2002). J. Mol. Biol. 315, 409–419. - PubMed
-
- Basselin, M., Coombs, G. H. & Barrett, M. P. (2000). Mol. Biochem. Parasitol. 109, 37–46. - PubMed
-
- Burchmore, R. J. & Hart, D. T. (1995). Mol. Biochem. Parasitol. 74, 77–86. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

