FGF21 is induced in cisplatin nephrotoxicity to protect against kidney tubular cell injury
- PMID: 29401620
- PMCID: PMC5956244
- DOI: 10.1096/fj.201701316R
FGF21 is induced in cisplatin nephrotoxicity to protect against kidney tubular cell injury
Abstract
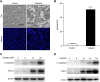
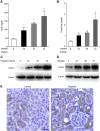
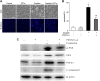
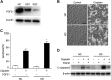
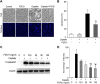
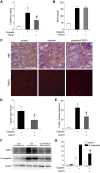
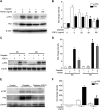
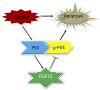
Cisplatin, a widely used cancer therapy drug, induces nephrotoxicity or acute kidney injury (AKI), but the underlying mechanism remains unclear, and renal protective approaches are not available. Fibroblast growth factor (FGF)21 is an endocrine factor that regulates glucose uptake, metabolism, and energy expenditure. However, recent work has also implicated FGF21 in cellular stress response under pathogenic conditions. The role and regulation of FGF21 in AKI are unclear. Here, we show that FGF21 was dramatically induced during cisplatin treatment of renal tubular cells in vitro and mouse kidneys in vivo. The inductive response was suppressed by pifithrin (a pharmacological inhibitor of P53), suggesting a role of P53 in FGF21 induction. In cultured renal tubular cells, knockdown of FGF21 aggravated cisplatin-induced apoptosis, whereas supplementation of recombinant FGF21 was protective. Consistently, recombinant FGF21 alleviated cisplatin-induced kidney dysfunction, tissue damage, and tubular apoptosis in mice. Mechanistically, FGF21 suppressed P53 induction and activation during cisplatin treatment. Together, these results indicate that FGF21 is induced during cisplatin nephrotoxicity to protect renal tubules, and recombinant FGF21 may have therapeutic potential.-Li, F., Liu, Z., Tang, C., Cai, J., Dong, Z. FGF21 is induced in cisplatin nephrotoxicity to protect against kidney tubular cell injury.
Keywords: P53; acute kidney injury; apoptosis.
Conflict of interest statement
The authors thank Dr. Jinke Cheng (Shanghai Jiao Tong University School of Medicine, Shanghai, China) for providing the FGF21 shRNA plasmids. This work was supported by the National Natural Science Foundation of China (Grants 81720108008 and 81430017), by the U.S. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases (Grants DK58831, DK87843), and by the U.S. Department of Veterans Affairs. The authors declare no conflicts of interest.
Figures
References
-
- Basile D. P., Bonventre J. V., Mehta R., Nangaku M., Unwin R., Rosner M. H., Kellum J. A., Ronco C.; ADQI XIII Work Group . (2016) Progression after AKI: understanding maladaptive repair processes to predict and identify therapeutic treatments. J. Am. Soc. Nephrol. 27, 687–697 10.1681/ASN.2015030309 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

