18F-VC701-PET and MRI in the in vivo neuroinflammation assessment of a mouse model of multiple sclerosis
- PMID: 29402285
- PMCID: PMC5800080
- DOI: 10.1186/s12974-017-1044-x
18F-VC701-PET and MRI in the in vivo neuroinflammation assessment of a mouse model of multiple sclerosis
Abstract
Background: Positron emission tomography (PET) using translocator protein (TSPO) ligands has been used to detect neuroinflammatory processes in neurological disorders, including multiple sclerosis (MS). The aim of this study was to evaluate neuroinflammation in a mouse MS model (EAE) using TSPO-PET with 18F-VC701, in combination with magnetic resonance imaging (MRI).
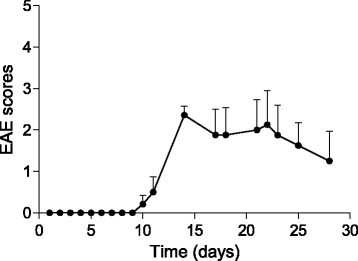
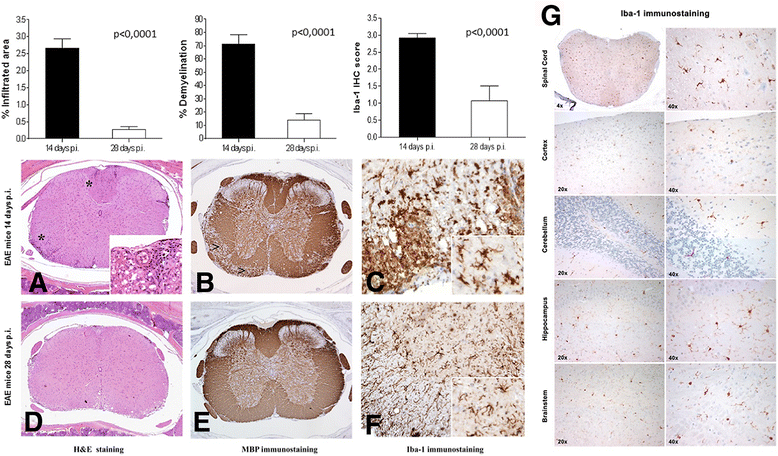
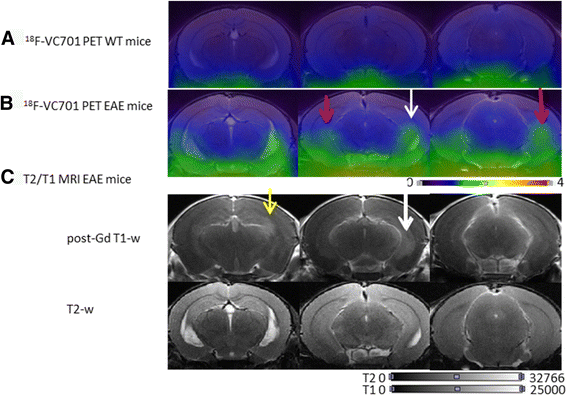
Methods: MOG35-55/CFA and pertussis toxin protocol was used to induce EAE in C57BL/6 mice. Disease progression was monitored daily, whereas MRI evaluation was performed at 1, 2, and 4 weeks post-induction. Microglia activation was assessed in vivo by 18F-VC701 PET at the time of maximum disease score and validated by radioligand ex vivo distribution and immunohistochemistry at 2 and 4 weeks post-immunization.
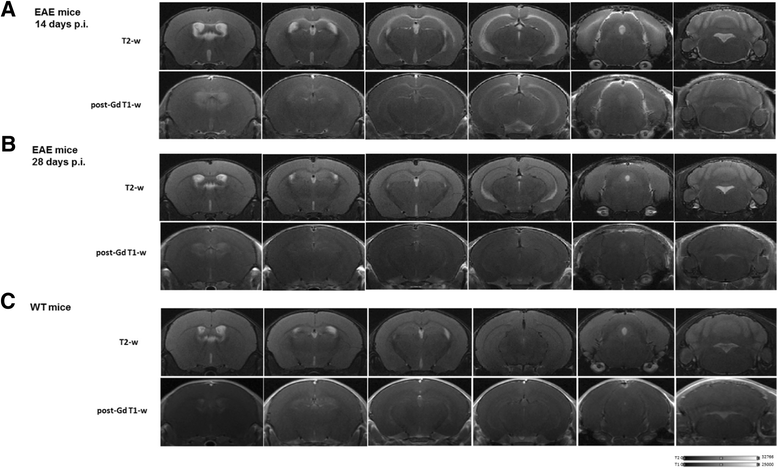
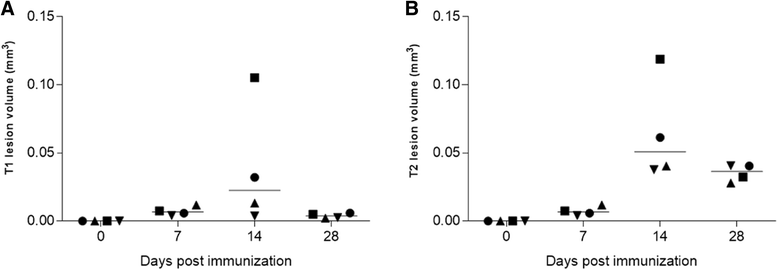
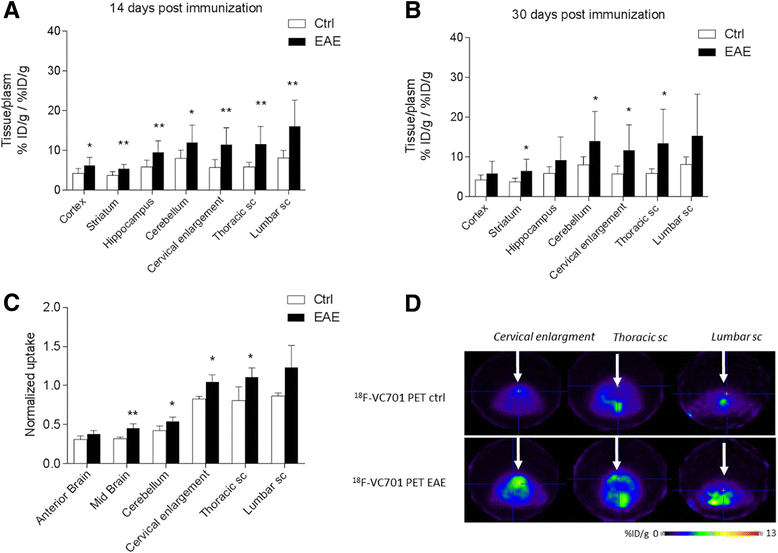
Results: In vivo and ex vivo analyses show that 18F-VC701 significantly accumulates within the central nervous system (CNS), particularly in the cortex, striatum, hippocampus, cerebellum, and cervical spinal cord of EAE compared to control mice, at 2 weeks post-immunization. MRI confirmed the presence of focal brain lesions at 2 weeks post-immunization in both T1-weighted and T2 images. Of note, MRI abnormalities attenuated in later post-immunization phase. Neuropathological analysis confirmed the presence of microglial activation in EAE mice, consistent with the in vivo increase of 18F-VC701 uptake.
Conclusion: Increase of 18F-VC701 uptake in EAE mice is strongly associated with the presence of microglia activation in the acute phase of the disease. The combined use of TSPO-PET and MRI provided complementary evidence on the ongoing disease process, thus representing an attractive new tool to investigate neuronal damage and neuroinflammation at preclinical levels.
Keywords: EAE monophasic model; MRI; Multiple sclerosis; Neuroinflammation; TSPO-PET.
Conflict of interest statement
Ethics approval
Animals were maintained and handled in compliance with the institutional guidelines for the care and use of experimental animals (IACUC), which have been notified to the Italian Ministry of Health and approved by the Ethics Committee of the San Raffaele Scientific Institute for studies involving animals.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing of interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
-
- Rasmussen S, Parsons AJ, Bassett S, Christensen MJ, Hume DE, Johnson LJ, Johnson RD, Simpson WR, Stacke C, Voisey CR, et al. High nitrogen supply and carbohydrate content reduce fungal endophyte and alkaloid concentration in Lolium perenne. New Phytol. 2007;173:787–797. doi: 10.1111/j.1469-8137.2006.01960.x. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

