Stepped-wedge randomised trial of laparoscopic ventral mesh rectopexy in adults with chronic constipation: study protocol for a randomized controlled trial
- PMID: 29402303
- PMCID: PMC5800022
- DOI: 10.1186/s13063-018-2456-3
Stepped-wedge randomised trial of laparoscopic ventral mesh rectopexy in adults with chronic constipation: study protocol for a randomized controlled trial
Abstract
Background: Laparoscopic ventral mesh rectopexy (LVMR) is an established treatment for external full-thickness rectal prolapse. However, its clinical efficacy in patients with internal prolapse is uncertain due to the lack of high-quality evidence.
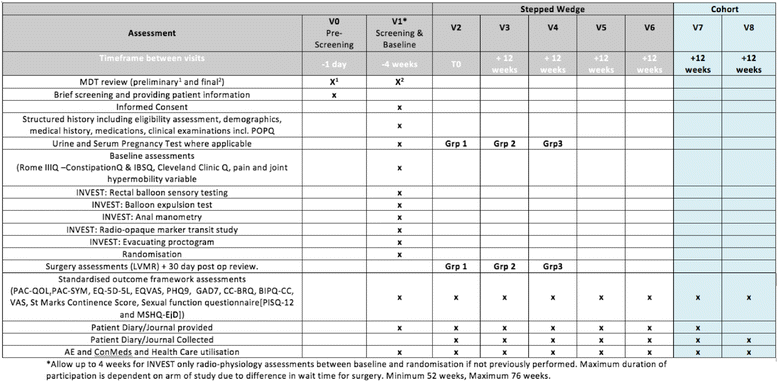
Methods: An individual level, stepped-wedge randomised trial has been designed to allow observer-blinded data comparisons between patients awaiting LVMR with those who have undergone surgery. Adults with symptomatic internal rectal prolapse, unresponsive to prior conservative management, will be eligible to participate. They will be randomised to three arms with different delays before surgery (0, 12 and 24 weeks). Efficacy outcome data will be collected at equally stepped time points (12, 24, 36 and 48 weeks). The primary objective is to determine clinical efficacy of LVMR compared to controls with reduction in the Patient Assessment of Constipation Quality of Life (PAC-QOL) at 24 weeks serving as the primary outcome. Secondary objectives are to determine: (1) the clinical effectiveness of LVMR to 48 weeks to a maximum of 72 weeks; (2) pre-operative determinants of outcome; (3) relevant health economics for LVMR; (4) qualitative evaluation of patient and health professional experience of LVMR and (5) 30-day morbidity and mortality rates.
Discussion: An individual-level, stepped-wedge, randomised trial serves the purpose of providing an untreated comparison for the active treatment group, while at the same time allowing the waiting-listed participants an opportunity to obtain the intervention at a later date. In keeping with the basic ethical tenets of this design, the average waiting time for LVMR (12 weeks) will be shorter than that for routine services (24 weeks).
Trial registration: ISRCTN registry, ISRCTN11747152 . Registered on 30 September 2015. The trial was prospectively registered (first patient enrolled on 21 March 2016).
Keywords: CapaCiTY; CapaCiTY study 3; Chronic constipation; Internal rectal prolapse; Laparoscopic ventral mesh rectopexy (LVMR); Rectopexy; Stepped wedge; Surgery.
Conflict of interest statement
Ethics approval and consent to participate
• Trial Registration: ISRCTN registry; registration number: 11747152.
• Research Ethics Committee (REC): London – City and East; REC reference number: 15/LO/0609. There is one main ethical review for all participating sites.
• Date of favourable ethical opinion: 6 July 2015.
• Substantial amendment date of favourable ethical opinion: 14 March 2016.
• Informed consent procedures: written informed consent will be obtained from research participants at visit 1 by an appropriately trained and delegated researcher. This will be done in a face-to-face setting in the clinic or private research environment.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing of interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

