Embryonic stem cell-derived cardiomyocytes for the treatment of doxorubicin-induced cardiomyopathy
- PMID: 29402309
- PMCID: PMC5799903
- DOI: 10.1186/s13287-018-0788-2
Embryonic stem cell-derived cardiomyocytes for the treatment of doxorubicin-induced cardiomyopathy
Abstract
Background: Doxorubicin (Dox) is a chemotherapy drug with limited application due to cardiotoxicity that may progress to heart failure. This study aims to evaluate the role of cardiomyocytes derived from mouse embryonic stem cells (CM-mESCs) in the treatment of Dox-induced cardiomyopathy (DIC) in mice.
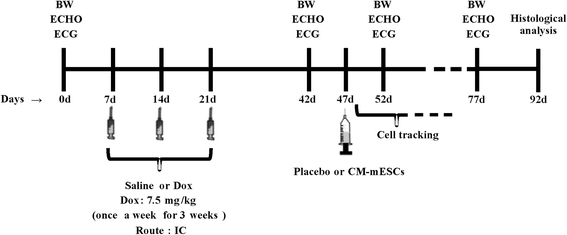
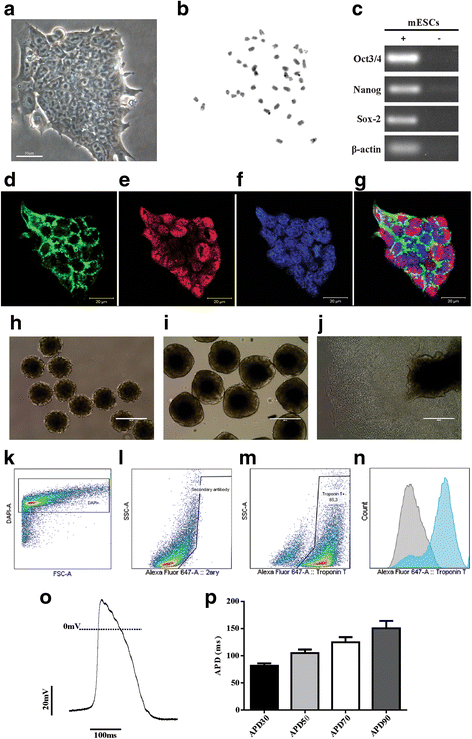
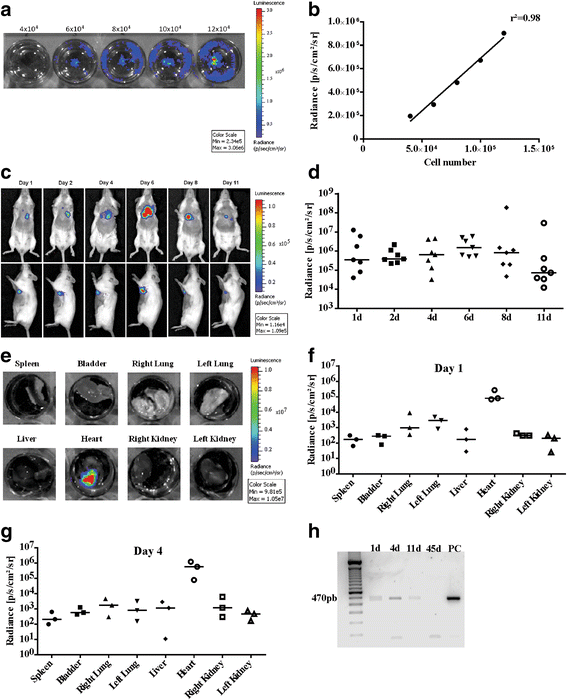
Methods: The mouse embryonic stem cell (mESC) line E14TG2A was characterized by karyotype analysis, gene expression using RT-PCR and immunofluorescence. Cells were transduced with luciferase 2 and submitted to cardiac differentiation. Total conditioned medium (TCM) from the CM-mESCs was collected for proteomic analysis. To establish DIC in CD1 mice, Dox (7.5 mg/kg) was administered once a week for 3 weeks, resulting in a cumulative Dox dose of 22.5 mg/kg. At the fourth week, a group of animals was injected intramyocardially with CM-mESCs (8 × 105 cells). Cells were tracked by a bioluminescence assay, and the body weight, echocardiogram, electrocardiogram and number of apoptotic cardiomyocytes were evaluated.
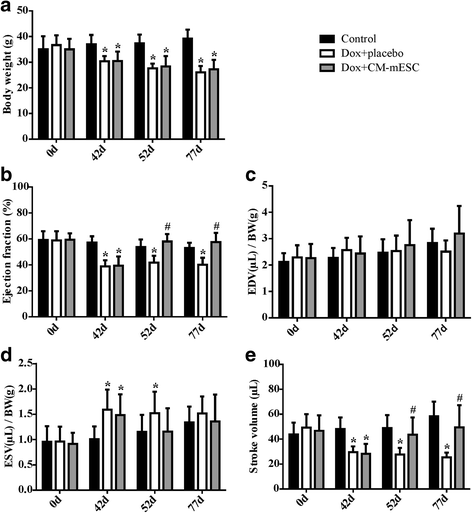
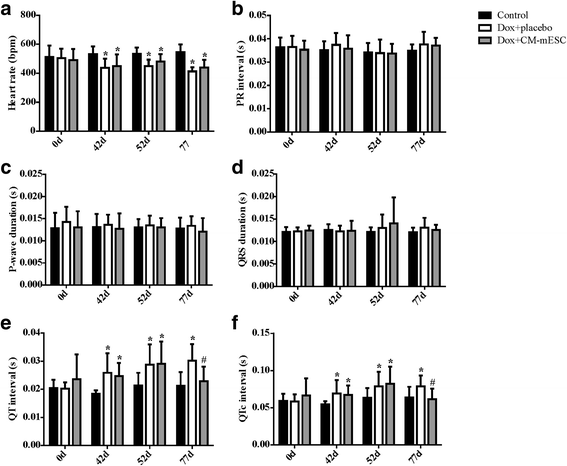
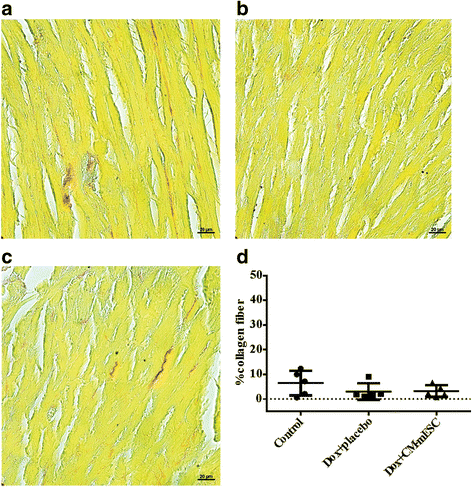
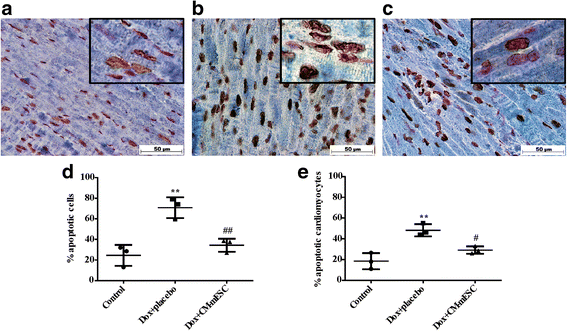
Results: mESCs exhibited a normal karyotype and expressed pluripotent markers. Proteomic analysis of TCM showed proteins related to the negative regulation of cell death. CM-mESCs presented ventricular action potential characteristics. Mice that received Dox developed heart failure and showed significant differences in body weight, ejection fraction (EF), end-systolic volume (ESV), stroke volume (SV), heart rate and QT and corrected QT (QTc) intervals when compared to the control group. After cell or placebo injection, the Dox + CM-mESC group showed significant increases in EF and SV when compared to the Dox + placebo group. Reduction in ESV and QT and QTc intervals in Dox + CM-mESC-treated mice was observed at 5 or 30 days after cell treatment. Cells were detected up to 11 days after injection. The Dox + CM-mESC group showed a significant reduction in the percentage of apoptotic cardiomyocytes in the hearts of mice when compared to the Dox + placebo group.
Conclusions: CM-mESC transplantation improves cardiac function in mice with DIC.
Keywords: Cardiomyocytes derived from mouse embryonic stem cells; Cell therapy; Dox-induced cardiomyopathy; Heart failure.
Conflict of interest statement
Ethics approval and consent to participate
This study was approved by the Ethics Committee for Animal Use of the Federal University of Rio de Janeiro under number IBCCF 171/13.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

