Analysis of species-dependent hydrolysis and protein binding of esmolol enantiomers
- PMID: 29403746
- PMCID: PMC5760893
- DOI: 10.1016/j.jpha.2012.01.007
Analysis of species-dependent hydrolysis and protein binding of esmolol enantiomers
Abstract
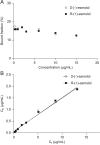
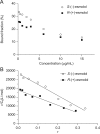
The stereoselective hydrolysis of esmolol in whole blood and in its separated components from rat, rabbit and human was investigated. Blood esterase activities were variable in different species in the order of rat>rabbit>human. Rat plasma showed the high esterase activity and had no stereoselectivity to enantiomers. Rabbit red blood cell (RBC) membrane, RBC cytosol and plasma all hydrolyzed esmolol but with different esterase activity, whereas the hydrolysis in RBC membrane and cytosol showed significant stereoselectivity towards R-(+)-esmolol. Esterase in RBC cytosol from human blood mainly contributed to the esmolol hydrolysis, which was demonstrated with no stereoselctivity. Esterase in human plasma showed a low activity, but a remarkable stereoselectivity with R-(+)-esmolol. In addition, the protein concentration affected the hydrolysis behavior of esmolol in RBC suspension. Protein binding of esmolol enantiomers in human plasma, human serum albumin (HSA) and α1-acid glycoprotein (AGP) revealed that there was a significant difference in bound fractions between two enantiomers, especially for AGP. Our results indicated that the stereoselective protein binding might play a role in the different hydrolysis rates of esmolol enantiomers in human plasma.
Keywords: Esmolol enantiomers; Protein binding; Species-dependent; Stereoselective hydrolysis.
Figures

Similar articles
-
Biochemical properties of blood esmolol esterase.Drug Metab Dispos. 1985 Jul-Aug;13(4):420-4. Drug Metab Dispos. 1985. PMID: 2863104
-
Species differences in the stereoselective hydrolysis of esmolol by blood esterases.Drug Metab Dispos. 1988 May-Jun;16(3):425-8. Drug Metab Dispos. 1988. PMID: 2900735
-
Esterases Involved in the Rapid Bioconversion of Esmolol after Intravenous Injection in Humans.Biol Pharm Bull. 2022;45(10):1544-1552. doi: 10.1248/bpb.b22-00468. Biol Pharm Bull. 2022. PMID: 36184514
-
Stereoselectivity in cutaneous hydrolysis and transdermal transport of propranolol prodrug.Enantiomer. 1997;2(3-4):181-91. Enantiomer. 1997. PMID: 9676267 Review.
-
In vitro, in silico and integrated strategies for the estimation of plasma protein binding. A review.Adv Drug Deliv Rev. 2015 Jun 23;86:27-45. doi: 10.1016/j.addr.2015.03.011. Epub 2015 Mar 27. Adv Drug Deliv Rev. 2015. PMID: 25819487 Review.
Cited by
-
Application of a UPLC-MS/MS method to the protein binding study of TM-2 in rat, human and beagle dog plasma.J Pharm Anal. 2016 Feb;6(1):32-38. doi: 10.1016/j.jpha.2015.08.001. Epub 2015 Aug 12. J Pharm Anal. 2016. PMID: 29403960 Free PMC article.
-
LC-MS/MS assay for the determination of natamycin in rabbit and human plasma: Application to a pharmacokinetics and protein binding study.J Pharm Anal. 2013 Apr;3(2):144-148. doi: 10.1016/j.jpha.2012.11.003. Epub 2012 Dec 12. J Pharm Anal. 2013. PMID: 29403809 Free PMC article.
-
Stereoselective binding of doxazosin enantiomers to plasma proteins from rats, dogs and humans in vitro.Acta Pharmacol Sin. 2013 Dec;34(12):1568-74. doi: 10.1038/aps.2013.120. Epub 2013 Nov 18. Acta Pharmacol Sin. 2013. PMID: 24241343 Free PMC article.
-
Fluorescent Probe as Dual-Organelle Localizer Through Differential Proton Gradients Between Lipid Droplets and Mitochondria.Anal Chem. 2024 Jun 4;96(22):9262-9269. doi: 10.1021/acs.analchem.4c01703. Epub 2024 May 17. Anal Chem. 2024. PMID: 38760019 Free PMC article.
References
-
- Van Besouw J. The use of short-acting beta-blockers in cardiac surgery. Esp. Anestesiol. Reanim. 2001;48(10):482–648. - PubMed
-
- Malovaná S., Gajdosová D., Benedík J. Determination of esmolol in serum by capillary zone electrophoresis and its monitoring in course of heart surgery. J. Chromatogr. B: Biomed. Sci. Appl. 2001;760(1):37–43. - PubMed
-
- Ariëns E.J., Wuis E.W. Bias in pharmacokinetics and clinical pharmacology. Clin. Pharmacol. Ther. 1987;42(4):361–363. - PubMed
-
- Nathanson J.A. Stereospecificity of beta adrenergic antagonists: R-enantiomers show increased selectivity for beta-2 receptors in ciliary process. Pharmacol. Exp. Ther. 1988;245(1):94–101. - PubMed
-
- Mehvar R., Brocks D.R. Impact of stereoselectivity on the pharmacokinetics and pharmacodynamics of antiarrhythmic drugs. J Pharm. Pharmaceut. Sci. 2001;4(2):185–200. - PubMed
LinkOut - more resources
Full Text Sources

