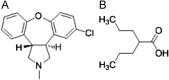
Simultaneous determination of asenapine and valproic acid in human plasma using LC-MS/MS: Application of the method to support pharmacokinetic study
- PMID: 29403845
- PMCID: PMC5761000
- DOI: 10.1016/j.jpha.2013.04.008
Simultaneous determination of asenapine and valproic acid in human plasma using LC-MS/MS: Application of the method to support pharmacokinetic study
Abstract
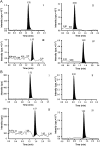
Combination of asenapine with valproic acid received regulatory approval for acute treatment of schizophrenia and maniac episodes of bipolar disorders. A simple LC-MS/MS method was developed and validated for simultaneous quantification of asenapine and valproic acid in human plasma. Internal standards were added to 300 μL of plasma sample prior to liquid-liquid extraction using methyl tertiary butyl ether (MTBE). Chromatographic separation was achieved on Phenomenex C18 column (50 mm×4.6 mm, 5 μm) in isocratic mode at 40 °C. The mobile phase used was 10 mM ammonium formate-acetonitrile (5:95, v/v) at a constant flow rate of 0.8 mL/min monitored on triple quadrupole mass spectrometer, operating in the multiple reaction monitoring (MRM) mode. The injection volume used for LC-MS/MS analysis was 15 μL and the run time was 2.5 min. These low run time and small injection volume suggest the high efficiency of the proposed method. The method was validated over the concentration range of 0.1-10.02 ng/mL and 10-20,000 ng/mL for asenapine and valproic acid respectively. The method recoveries of asenapine (81.33%), valproic acid (81.70%), gliclazide (78.45%) and benzoic acid (79.73) from spiked plasma samples were consistent and reproducible. The application of this method was demonstrated by a pharmacokinetic study in 8 healthy male volunteers with 5 mg asenapine and 250 mg valproic acid administration.
Keywords: Asenapine; Bipolar disorders; Gliclazide; Pharmacokinetics; Schizophrenia; Valproic acid.
Figures


References
-
- McIntyre R.S., Muzina D.J., Kemp D.E. Bipolar disorder and suicide: research synthesis and clinical translation. Curr. Psychiatry Rep. 2008;10(1):66–72. - PubMed
-
- Schneck C.D., Miklowitz D.J., Miyahara S. The prospective course of rapid-cycling bipolar disorder: findings from the STEP-BD. Am. J. Psychiatry. 2008;165(3):370–377. - PubMed
-
- Costall B., Domeney A.M., Kelly M.E. Actions of ORG 5222 as a novel psychotropic agent. Pharmacol. Biochem. Behav. 1990;35(3):607–615. - PubMed
-
- Krishnan K.R. Psychiatric and medical comorbidities of bipolar disorder. Psychosom. Med. 2005;67(1):1–8. - PubMed
-
- Baldessarini R.J., Vieta E., Calabrese J.R. Bipolar depression: overview and commentary. Harv. Rev. Psychiatry. 2010;18(3):143–157. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

