Quality evaluation of synthetic quorum sensing peptides used in R&D
- PMID: 29403929
- PMCID: PMC5762210
- DOI: 10.1016/j.jpha.2014.12.002
Quality evaluation of synthetic quorum sensing peptides used in R&D
Abstract
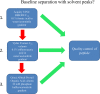
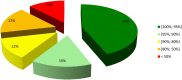
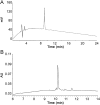
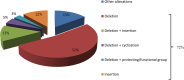
Peptides are becoming an important class of molecules in the pharmaceutical field. Closely related peptide-impurities in peptides are inherent to the synthesis approach and have demonstrated to potentially mask biomedical experimental results. Quorum sensing peptides are attracting high interest in R&D and therefore a representative set of quorum sensing peptides, with a requested purity of at least 95.0%, was evaluated for their purity and nature of related impurities. In-house quality control (QC) revealed a large discrepancy between the purity levels as stated on the supplier׳s certificate of analysis and our QC results. By using our QC analysis flowchart, we demonstrated that only 44.0% of the peptides met the required purity. The main compound of one sample was even found to have a different structure compared to the desired peptide. We also found that the majority of the related impurities were lacking amino acid(s) in the desired peptide sequence. Relying on the certificates of analysis as provided by the supplier might have serious consequences for peptide research, and peptide-researchers should implement and maintain a thorough in-house QC.
Keywords: Impurity profiling; Quality; Quorum sensing peptides.
Figures





Similar articles
-
Quality control of cationic cell-penetrating peptides.J Pharm Biomed Anal. 2016 Jan 5;117:289-97. doi: 10.1016/j.jpba.2015.09.011. Epub 2015 Sep 12. J Pharm Biomed Anal. 2016. PMID: 26397208
-
Enantiomeric purity of synthetic therapeutic peptides: A review.Chirality. 2024 Mar;36(3). doi: 10.1002/chir.23652. Chirality. 2024. PMID: 38448043 Review.
-
Structure-function analysis of peptide signaling in the Clostridium perfringens Agr-like quorum sensing system.J Bacteriol. 2015 May;197(10):1807-18. doi: 10.1128/JB.02614-14. Epub 2015 Mar 16. J Bacteriol. 2015. PMID: 25777675 Free PMC article.
-
Structure-activity analysis of quorum-sensing signaling peptides from Streptococcus mutans.J Bacteriol. 2007 Feb;189(4):1441-50. doi: 10.1128/JB.00832-06. Epub 2006 Aug 25. J Bacteriol. 2007. PMID: 16936029 Free PMC article.
-
Related impurities in peptide medicines.J Pharm Biomed Anal. 2014 Dec;101:2-30. doi: 10.1016/j.jpba.2014.06.012. Epub 2014 Jun 13. J Pharm Biomed Anal. 2014. PMID: 25044089 Review.
Cited by
-
Quantification of a peptide standard using the intrinsic fluorescence of tyrosine.Anal Bioanal Chem. 2016 Mar;408(9):2187-93. doi: 10.1007/s00216-016-9334-1. Epub 2016 Feb 15. Anal Bioanal Chem. 2016. PMID: 26879647 Free PMC article.
-
Enfuvirtide biosynthesis in thermostable chaperone-based fusion.Biotechnol Rep (Amst). 2022 May 14;35:e00734. doi: 10.1016/j.btre.2022.e00734. eCollection 2022 Sep. Biotechnol Rep (Amst). 2022. PMID: 35646620 Free PMC article.
-
Quorum Sensing Peptides Selectively Penetrate the Blood-Brain Barrier.PLoS One. 2015 Nov 4;10(11):e0142071. doi: 10.1371/journal.pone.0142071. eCollection 2015. PLoS One. 2015. PMID: 26536593 Free PMC article.
-
Discovery of Melittin as Triple-Action Agent: Broad-Spectrum Antibacterial, Anti-Biofilm, and Potential Anti-Quorum Sensing Activities.Molecules. 2024 Jan 23;29(3):558. doi: 10.3390/molecules29030558. Molecules. 2024. PMID: 38338303 Free PMC article.
References
-
- Craik D., Fairlie D., Liras S. The future of peptide-based drugs. Chem. Biol. Drug Des. 2013;81:136–147. - PubMed
-
- Berggren R., Moller M., Moss R. Outlook for the next 5 years in drug innovation. Nat. Rev. Drug Discov. 2012;11:435–436. - PubMed
-
- Van Arnum P. Peptides gain traction in drug development. Pharm. Technol. 2012;36:42–43.
-
- Albericio F., Kruger H. Therapeutic peptides. Future Med. Chem. 2012;4:1527–1531. - PubMed
-
- Danho W., Swistok J., Khan W. Peptides for Youth. In: Del Valle S., Escher E., Lubell W., editors. Proceedings of the 20th American Peptide Symposium. Springer Sciences and Business Media; NewYork, NY, USA: 2009. pp. 467–468.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

