Identification, synthesis and characterization of process related impurities of benidipine hydrochloride, stress-testing/stability studies and HPLC/UPLC method validations
- PMID: 29403939
- PMCID: PMC5762216
- DOI: 10.1016/j.jpha.2015.02.001
Identification, synthesis and characterization of process related impurities of benidipine hydrochloride, stress-testing/stability studies and HPLC/UPLC method validations
Abstract
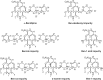
Benidipine hydrochloride, used as an antihypertensive agent and long-acting calcium antagonist, is synthesized for commercial use as a drug substance in highly pure form. During the synthetic process development studies of benidipine, process related impurities were detected. These impurities were identified, synthesized and characterized and mechanisms of their formation were discussed in detail. After all standardization procedures, they were used as reference standards for analytical studies. In addition, a separate HPLC method was developed and validated for detection of residual 1-benzylpiperidin-3-ol (Ben-2), which is used during benidipine synthesis and controlled as a potential process related impurity. As complementary of this work, stress-testing studies of benidipine were carried out under specified conditions and a stability-indicating UPLC assay method was developed, validated and used during stability studies of benidipine.
Keywords: Benidipine; Characterization; Impurities; Stability; Synthesis; Validation.
Figures








Similar articles
-
Identification, synthesis and characterization of process related desfluoro impurity of ezetimibe and HPLC method validations.J Pharm Anal. 2015 Dec;5(6):356-370. doi: 10.1016/j.jpha.2015.04.002. Epub 2015 Apr 24. J Pharm Anal. 2015. PMID: 29403950 Free PMC article.
-
Synthesis, characterization and control of eight process-related and two genotoxic fingolimod impurities by a validated RP-UPLC method.Biomed Chromatogr. 2022 Apr;36(4):e5316. doi: 10.1002/bmc.5316. Epub 2022 Jan 31. Biomed Chromatogr. 2022. PMID: 34981532
-
Development of RP UPLC-TOF/MS, stability indicating method for omeprazole and its related substances by applying two level factorial design; and identification and synthesis of non-pharmacopoeial impurities.J Pharm Biomed Anal. 2016 Jan 25;118:370-379. doi: 10.1016/j.jpba.2015.10.005. Epub 2015 Nov 21. J Pharm Biomed Anal. 2016. PMID: 26600119
-
Pharmacological, pharmacokinetic, and clinical properties of benidipine hydrochloride, a novel, long-acting calcium channel blocker.J Pharmacol Sci. 2006 Apr;100(4):243-61. doi: 10.1254/jphs.dtj05001x. Epub 2006 Mar 25. J Pharmacol Sci. 2006. PMID: 16565579 Review.
-
Development of Impurity Profiling Methods Using Modern Analytical Techniques.Crit Rev Anal Chem. 2017 Jan 2;47(1):24-36. doi: 10.1080/10408347.2016.1169913. Epub 2016 Apr 12. Crit Rev Anal Chem. 2017. PMID: 27070830 Review.
Cited by
-
Identification, synthesis and characterization of process related desfluoro impurity of ezetimibe and HPLC method validations.J Pharm Anal. 2015 Dec;5(6):356-370. doi: 10.1016/j.jpha.2015.04.002. Epub 2015 Apr 24. J Pharm Anal. 2015. PMID: 29403950 Free PMC article.
References
-
- Yao K., Nagashima K., Miki H. Pharmacological, pharmacokinetic, and clinical properties of benidipine hydrochloride, a novel, long-acting calcium channel blocker. J. Pharmacol. Sci. 2006;100:243–261. - PubMed
-
- Takayama M., Matsubara M., Arakawa E., et al. Comparison of the antiatherosclerotic effects of dihydropyridine calcium channel blocker and HMG-CoA reductase inhibitor on hypercholesterolemic rabbits. Vasc. Pharmacol. 2007;46:302–308. - PubMed
-
- Suzuki H., Ono E., Ueno H., et al. Physico-chemical properties and stabilities of the highly potent calcium antagonist benidipine hydrochloride. Arzneim. Forsch./Drug Res. 1988;38:1671–1676. - PubMed
-
- Prous J., Castaner J. KW-3049. Drugs Future. 1986;11:936–938.
-
- Kalyanaramu B., Raghubabu K. Development of new visible spectrophotometric methods for determination of benidipine hydrochloride in bulk and formulations based on oxidative coupling and diazo coupling reactions. Int. J. Pharm. Biol. Sci. 2011;1:57–66.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

