Broad anti-herpesviral activity of α-hydroxytropolones
- PMID: 29408023
- PMCID: PMC6292515
- DOI: 10.1016/j.vetmic.2017.12.016
Broad anti-herpesviral activity of α-hydroxytropolones
Abstract
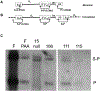
Herpesviruses are ubiquitous in animals and cause economic losses concomitant with many diseases. Most of the domestic animal herpesviruses are within the subfamily Alphaherpesvirinae, which includes human herpes simplex virus 1 (HSV-1). Suppression of HSV-1 replication has been reported with α-hydroxytropolones (αHTs), aromatic ring compounds that have broad bioactivity due to potent chelating activity. It is postulated that αHTs inhibit enzymes within the nucleotidyltransferase superfamily (NTS). These enzymes require divalent cations for nucleic acid cleavage activity. Potential targets include the nuclease component of the herpesvirus terminase (pUL15C), a highly conserved NTS-like enzyme that cleaves viral DNA into genomic lengths prior to packaging into capsids. Inhibition of pUL15C activity in biochemical assays by various αHTs previously revealed a spectrum of potencies. Interestingly, the most potent anti-pUL15C αHT inhibited HSV-1 replication to a limited extent in cell culture. The aim of this study was to evaluate three different αHT molecules with varying biochemical anti-pUL15C activity for a capacity to inhibit replication of veterinary herpesviruses (BoHV-1, EHV-1, and FHV-1) and HSV-1. Given the known discordant potencies between anti-pUL15C and HSV-1 replication inhibition, a second objective was to elucidate the mechanism of action of these compounds. The results show that αHTs broadly inhibit herpesviruses, with similar inhibitory effect against HSV-1, BoHV-1, EHV-1, and FHV-1. Based on immunoblotting, Southern blotting, and real-time qPCR, the compounds were found to specifically inhibit viral DNA replication. Thus, αHTs represent a new class of broadly active anti-herpesviral compounds with potential veterinary applications.
Keywords: Anti-herpesvirus; Herpesvirus; Nucleotidyltransferase superfamily; α-hydroxytropolones.
Copyright © 2017 Elsevier B.V. All rights reserved.
Figures
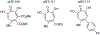



References
-
- Bentley R, 2008. A fresh look at natural tropolonoids. Nat. Prod. Rep 25, 118–138. - PubMed
-
- Biswas S, Bandyopadhyay S, Dimri U, Patra PH, 2013. Bovine herpesvirus-1 (BHV-1) – a re-emerging concern in livestock: a revisit to its biology, epidemiology, diagnosis, and prophylaxis. Vet. Q 33, 68–81. - PubMed
-
- Boehmer PE, Lehman IR, 1997. Herpes simplex virus DNA replication. Annu. Rev. Biochem 66, 347–384. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

