Optimization of commercially available Zika virus antibodies for use in a laboratory-developed immunohistochemical assay
- PMID: 29416874
- PMCID: PMC5783976
- DOI: 10.1002/cjp2.84
Optimization of commercially available Zika virus antibodies for use in a laboratory-developed immunohistochemical assay
Abstract
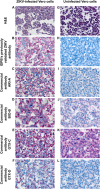
Zika virus (ZIKV) infection during pregnancy can cause adverse fetal outcomes and severe irreversible congenital birth defects including microcephaly. Immunohistochemistry (IHC) is a valuable diagnostic tool for detecting ZIKV antigens in tissues from cases of fetal loss in women infected with ZIKV, and for providing insights into disease pathogenesis. As a result, there is increasing demand for commercially available ZIKV antibodies for use in IHC assays. ZIKV antibodies were selected and obtained from commercial sources to include both mouse and rabbit hosts, and a variety of antigenic targets. Pretreatment conditions and antibody concentrations resulting in optimal immunohistochemical staining were determined using ZIKV cell control and polymerase chain reaction (PCR)-confirmed ZIKV case control material (fetal brain tissue). Cross-reactivity of the antibodies against other flaviviruses (dengue virus serogroups 1-4, yellow fever virus, Japanese encephalitis virus, West Nile virus) and chikungunya virus was also evaluated. Immunostaining using the commercially available antibodies was compared to a previously validated ZIKV IHC assay used for primary diagnosis. Four antibodies demonstrated optimal staining similar to the previously validated ZIKV IHC assay. Two of the four antibodies cross-reacted with dengue virus, while the other two antibodies showed no cross-reactivity with dengue, other flaviviruses, or chikungunya virus. Differences in the cross-reactivity profiles could not be entirely explained by the antigenic target. Commercially available ZIKV antibodies can be optimized for use in IHC testing to aid in ZIKV diagnostic testing and an evaluation of tissue tropism.
Keywords: Zika virus; antibody; fetal demise; immunohistochemistry; microcephaly; pathology; placenta.
Figures

References
-
- Gould EA, Solomon T. Pathogenic flaviviruses. Lancet 2008; 371 : 500–509. - PubMed
-
- Duffy MR, Chen TH, Hancock WT, et al Zika virus outbreak on Yap Island, Federated States of Micronesia. N Engl J Med 2009; 360 : 2536–2543. - PubMed
-
- Rasmussen SA, Jamieson DJ, Honein MA, et al Zika virus and birth defects–reviewing the evidence for causality. N Engl J Med 2016; 374 : 1981–1987. - PubMed
-
- Rubin EJ, Greene MF, Baden LR. Zika virus and microcephaly. N Engl J Med 2016; 374 : 984–985. - PubMed
-
- Ritter JM, Martines RB, Zaki SR. Zika virus: pathology from the pandemic. Arch Pathol Lab Med 2017; 141 : 49–59. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

