Motor neuron degeneration correlates with respiratory dysfunction in SCA1
- PMID: 29419414
- PMCID: PMC5894948
- DOI: 10.1242/dmm.032623
Motor neuron degeneration correlates with respiratory dysfunction in SCA1
Abstract
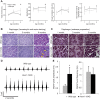
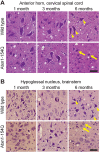
Spinocerebellar ataxia type 1 (SCA1) is characterized by adult-onset cerebellar degeneration with attendant loss of motor coordination. Bulbar function is eventually impaired and patients typically die from an inability to clear the airway. We investigated whether motor neuron degeneration is at the root of bulbar dysfunction by studying SCA1 knock-in (Atxn1154Q/+ ) mice. Spinal cord and brainstem motor neurons were assessed in Atxn1154Q/+ mice at 1, 3 and 6 months of age. Specifically, we assessed breathing physiology, diaphragm histology and electromyography, and motor neuron histology and immunohistochemistry. Atxn1154Q/+ mice show progressive neuromuscular respiratory abnormalities, neurogenic changes in the diaphragm, and motor neuron degeneration in the spinal cord and brainstem. Motor neuron degeneration is accompanied by reactive astrocytosis and accumulation of Atxn1 aggregates in the motor neuron nuclei. This observation correlates with previous findings in SCA1 patient tissue. Atxn1154Q/+ mice develop bulbar dysfunction because of motor neuron degeneration. These findings confirm the Atxn1154Q/+ line as a SCA1 model with face and construct validity for this understudied disease feature. Furthermore, this model is suitable for studying the pathogenic mechanism driving motor neuron degeneration in SCA1 and possibly other degenerative motor neuron diseases. From a clinical standpoint, the data indicate that pulmonary function testing and employment of non-invasive ventilator support could be beneficial in SCA1 patients. The physiological tests used in this study might serve as valuable biomarkers for future therapeutic interventions and clinical trials.This article has an associated First Person interview with the first author of the paper.
Keywords: Bulbar dysfunction; Motor neuron degeneration; SCA1; Spinocerebellar ataxia type 1.
© 2018. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

