Adiponectin aggravates bone erosion by promoting osteopontin production in synovial tissue of rheumatoid arthritis
- PMID: 29422077
- PMCID: PMC5806355
- DOI: 10.1186/s13075-018-1526-y
Adiponectin aggravates bone erosion by promoting osteopontin production in synovial tissue of rheumatoid arthritis
Erratum in
-
Correction to: Adiponectin aggravates bone erosion by promoting osteopontin production in synovial tissue of rheumatoid arthritis.Arthritis Res Ther. 2018 Jun 13;20(1):125. doi: 10.1186/s13075-018-1641-9. Arthritis Res Ther. 2018. PMID: 29898766 Free PMC article.
Abstract
Background: We have previously reported that adiponectin (AD), an adipokine that is secreted by adipocytes, correlates well with progressive bone erosion in rheumatoid arthritis (RA). The exact mechanism of AD in promoting joint destruction remains unclear. Osteopontin (OPN) is required for osteoclast recruitment. We hypothesized that AD exacerbates bone erosion by inducing OPN expression in synovial tissue. This study aimed to evaluate a novel role for AD in RA.
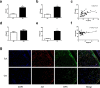
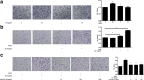
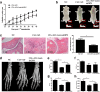
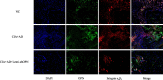
Methods: The serum levels of AD and OPN were determined in 38 patients with RA, 40 patients with osteoarthritis (OA), and 20 healthy controls using enzyme-linked immunosorbent assay (ELISA). AD and OPN production were measured by double immunofluorescence in RA and OA synovial tissue. Quantitative real-time PCR and immunofluorescence were used to evaluate the mRNA and protein expression levels of OPN in RA synovial fibroblasts (RASFs) and OA synovial fibroblasts after pre-incubation with AD, respectively. Migration of the RAW264.7 osteoclast precursor cell line was assessed using the Transwell migration assay and co-culture system. Bone destruction and osteoclastogenesis were assessed by immunohistochemical staining, microcomputed tomography and tartrate-resistant acid phosphatase (TRAP) staining in AD-treated collagen-induced arthritis (CIA) mice with or without OPN silencing. The expression levels of OPN and integrin αvβ3 in the ankle joint tissues of the mice were examined by double immunofluorescence.
Results: Our results indicated that the AD and OPN expression levels increased noticeably and were associated with each other in the RA serum. The AD distribution was coincident with that of OPN in the RA synovial tissue. AD stimulation of RASFs increased OPN production in a dose-dependent manner. AD-treated RASFs promoted RAW264.7 cell migration, and the effect was blocked with a specific antibody against OPN. Silencing of OPN using lentiviral-OPN short hairpin RNA reduced the number of TRAP-positive osteoclasts and the extent of bone erosion in the AD-treated CIA mice. When bound to integrin αvβ3, OPN functions as a mediator of AD and osteoclasts.
Conclusions: Our study provides new evidence of AD involvement in bone erosion. AD induces the expression of OPN, which recruits osteoclasts and initiates bone erosion. These data highlight AD as a novel target for RA treatment.
Keywords: Adiponectin; Bone; Erosion; Osteoclasts; Osteopontin; Rheumatoid arthritis.
Conflict of interest statement
Ethics approval and consent to participate
This study was approved by the Ethics Committee at the First Affiliated Hospital of Nanjing Medical University, and all donors signed written informed consent forms. The animal experiments were conducted in compliance with the guidelines for the care and use of laboratory animals and approved by Institutional Animal Care and Use Committee of Nanjing Medical University.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


Similar articles
-
Osteopontin inhibition of miR-129-3p enhances IL-17 expression and monocyte migration in rheumatoid arthritis.Biochim Biophys Acta Gen Subj. 2017 Feb;1861(2):15-22. doi: 10.1016/j.bbagen.2016.11.015. Epub 2016 Nov 13. Biochim Biophys Acta Gen Subj. 2017. PMID: 27851983
-
Increased amount of phosphorylated proinflammatory osteopontin in rheumatoid arthritis synovia is associated to decreased tartrate-resistant acid phosphatase 5B/5A ratio.PLoS One. 2017 Aug 8;12(8):e0182904. doi: 10.1371/journal.pone.0182904. eCollection 2017. PLoS One. 2017. PMID: 28792533 Free PMC article.
-
Adiponectin promotes fibroblast-like synoviocytes producing IL-6 to enhance T follicular helper cells response in rheumatoid arthritis.Clin Exp Rheumatol. 2020 Jan-Feb;38(1):11-18. Epub 2019 Apr 11. Clin Exp Rheumatol. 2020. PMID: 31025923
-
Role of osteopontin in rheumatoid arthritis.Rheumatol Int. 2015 Apr;35(4):589-95. doi: 10.1007/s00296-014-3122-z. Epub 2014 Aug 28. Rheumatol Int. 2015. PMID: 25163663 Review.
-
Osteopontin: A Bone-Derived Protein Involved in Rheumatoid Arthritis and Osteoarthritis Immunopathology.Biomolecules. 2023 Mar 9;13(3):502. doi: 10.3390/biom13030502. Biomolecules. 2023. PMID: 36979437 Free PMC article. Review.
Cited by
-
The Effect of a Comprehensive Life-Style Intervention Program of Diet and Exercise on Four Bone-Derived Proteins, FGF-23, Osteopontin, NGAL and Sclerostin, in Overweight or Obese Children and Adolescents.Nutrients. 2022 Sep 13;14(18):3772. doi: 10.3390/nu14183772. Nutrients. 2022. PMID: 36145151 Free PMC article.
-
Adipocytokines: Are they the Theory of Everything?Cytokine. 2020 Sep;133:155144. doi: 10.1016/j.cyto.2020.155144. Epub 2020 Jun 16. Cytokine. 2020. PMID: 32559663 Free PMC article. Review.
-
Adipokines in Rheumatoid Arthritis: Emerging Biomarkers and Therapeutic Targets.Biomedicines. 2023 Nov 8;11(11):2998. doi: 10.3390/biomedicines11112998. Biomedicines. 2023. PMID: 38001998 Free PMC article. Review.
-
The anti-diarrhea activity of red algae-originated sulphated polysaccharides on ETEC-K88 infected mice.RSC Adv. 2019 Jan 18;9(5):2360-2370. doi: 10.1039/c8ra09247h. eCollection 2019 Jan 18. RSC Adv. 2019. PMID: 35520502 Free PMC article.
-
Adipocytokines in Rheumatoid Arthritis: The Hidden Link between Inflammation and Cardiometabolic Comorbidities.J Immunol Res. 2018 Nov 21;2018:8410182. doi: 10.1155/2018/8410182. eCollection 2018. J Immunol Res. 2018. PMID: 30584543 Free PMC article. Review.
References
-
- Andersen M, Boesen M, Ellegaard K, Christensen R, Soderstrom K, Soe N, Spee P, Morch UG, Torp-Pedersen S, Bartels EM, et al. Synovial explant inflammatory mediator production corresponds to rheumatoid arthritis imaging hallmarks: a cross-sectional study. Arthritis Res Ther. 2014;16(3):R107. doi: 10.1186/ar4557. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

