Associations between fatty acid oxidation, hepatic mitochondrial function, and plasma acylcarnitine levels in mice
- PMID: 29422939
- PMCID: PMC5789604
- DOI: 10.1186/s12986-018-0241-7
Associations between fatty acid oxidation, hepatic mitochondrial function, and plasma acylcarnitine levels in mice
Abstract
Background: The 4-thia fatty acid tetradecylthiopropionic acid (TTP) is known to inhibit mitochondrial β-oxidation, and can be used as chemically induced hepatic steatosis-model in rodents, while 3-thia fatty acid tetradecylthioacetic acid (TTA) stimulates fatty acid oxidation through activation of peroxisome proliferator activated receptor alpha (PPARα). We wished to determine how these two compounds affected in vivo respiration and mitochondrial efficiency, with an additional goal to elucidate whether mitochondrial function is reflected in plasma acylcarnitine levels.
Methods: C57BL/6 mice were divided in 4 groups of 10 mice and fed a control low-fat diet, low-fat diets with 0.4% (w/w) TTP, 0.4% TTA or a combination of these two fatty acids for three weeks (n = 10). At sacrifice, β-oxidation and oxidative phosphorylation (OXPHOS) capacity was analysed in fresh liver samples. Hepatic mitochondria were studied using transmission electron microscopy. Lipid classes were measured in plasma, heart and liver, acylcarnitines were measured in plasma, and gene expression was measured in liver.
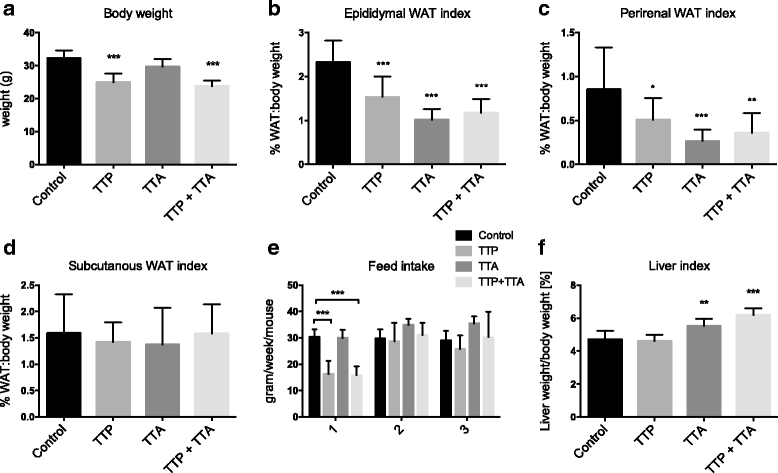
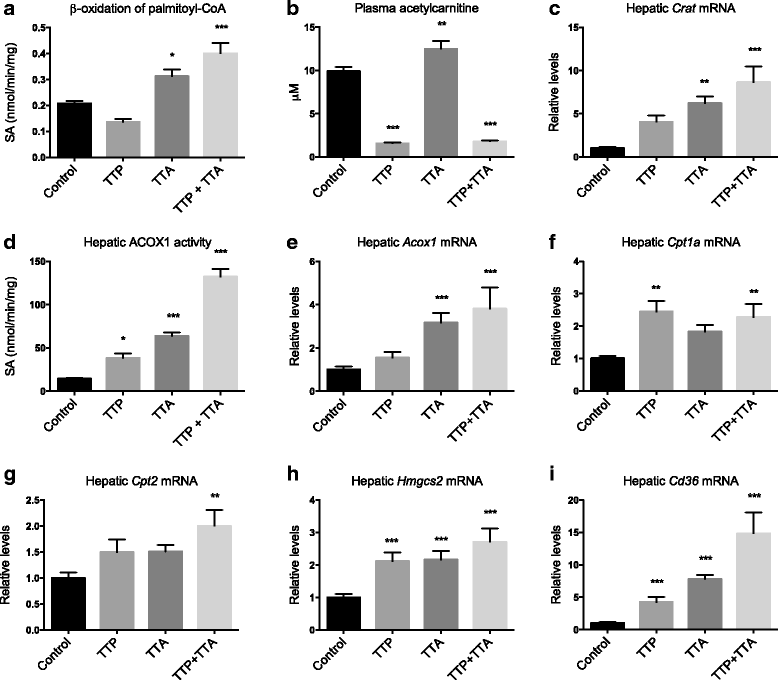
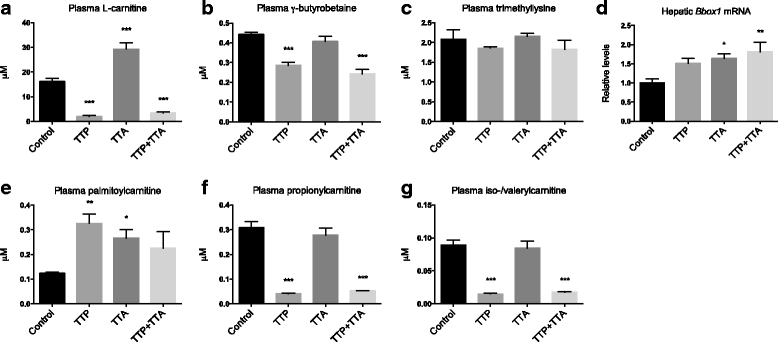
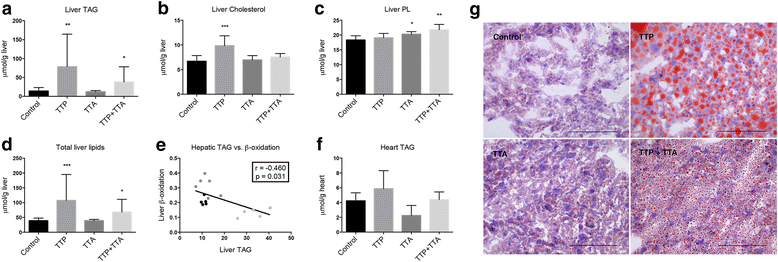
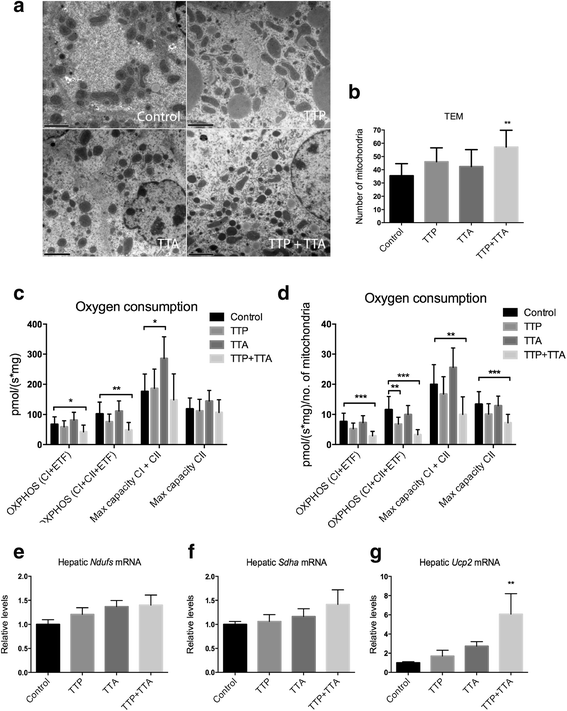
Results: The TTP diet resulted in hepatic lipid accumulation, plasma L-carnitine and acetylcarnitine depletion and elevated palmitoylcarnitine and non-esterified fatty acid levels. No significant lipid accumulation was observed in heart. The TTA supplement resulted in enhanced hepatic β-oxidation, accompanied by an increased level of acetylcarnitine and palmitoylcarnitine in plasma. Analysis of mitochondrial respiration showed that TTP reduced oxidative phosphorylation, while TTA increased the maximum respiratory capacity of the electron transport system. Combined treatment with TTP and TTA resulted in a profound stimulation of genes involved in the PPAR-response and L-carnitine metabolism, and partly prevented triacylglycerol accumulation in the liver concomitant with increased peroxisomal β-oxidation and depletion of plasma acetylcarnitines. Despite an increased number of mitochondria in the liver of TTA + TTP fed mice, the OXPHOS capacity was significantly reduced.
Conclusion: This study indicates that fatty acid β-oxidation directly affects mitochondrial respiratory capacity in liver. As plasma acylcarnitines reflected the reduced mitochondrial β-oxidation in TTP-fed mice, they could be useful tools to monitor mitochondrial function. As mitochondrial dysfunction is a major determinant of metabolic disease, this supports their use as plasma markers of cardiovascular risk in humans. Results however indicate that high PPAR activation obscures the interpretation of plasma acylcarnitine levels.
Keywords: Acylcarnitine; Beta-oxidation; Mitochondrial function; Oxidative phosphorylation; Tetradecylthioacetic acid; Tetradecylthiopropionic acid.
Conflict of interest statement
The animal study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals. The protocol was approved by the Norwegian State Board of Biological Experiments with Living Animals (Project no. 5071).Not applicableThe authors declare that they have no competing interests.Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

