Schisandrol B and schisandrin B inhibit TGFβ1-mediated NF-κB activation via a Smad-independent mechanism
- PMID: 29423034
- PMCID: PMC5790451
- DOI: 10.18632/oncotarget.23213
Schisandrol B and schisandrin B inhibit TGFβ1-mediated NF-κB activation via a Smad-independent mechanism
Abstract
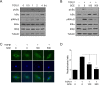
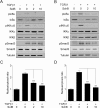
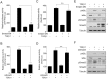
Aberrant transforming growth factor β1 (TGFβ1) signaling plays a pathogenic role in the development of vascular fibrosis. We have reported that Schisandra chinensis fruit extract (SCE), which has been used as a traditional oriental medicine, suppresses TGFβ1-mediated phenotypes in vascular smooth muscle cells (VSMCs). However, it is still largely unknown about the pharmacologic effects of SCE on various TGFβ1 signaling components. In this study, we found that SCE attenuated TGFβ1-induced NF-κB activation and nuclear translocation in VSMCs. Among the five active ingredients of SCE that were examined, schisandrol B (SolB) and schisandrin B (SchB) most potently suppressed TGFβ1-mediated NF-κB activation. In addition, SolB and SchB effectively inhibited IKKα/β activation and IκBα phosphorylation in TGFβ1-treated VSMCs. The pharmacologic effects of SolB and SchB on NF-κB activation were independent of the Smad-mediated canonical pathway. Therefore, our study demonstrates that SCE and its active constituents SolB and SchB suppress TGFβ1-mediated NF-κB signaling pathway in a Smad-independent mechanism. Our results may help further investigations to develop novel multi-targeted therapeutic strategies that treat or prevent vascular fibrotic diseases.
Keywords: NF-κB; TGFβ1; schisandra chinensis; schisandrin B; schisandrol B.
Conflict of interest statement
CONFLICTS OF INTEREST None declared.
Figures



Similar articles
-
Schisandrin B suppresses TGFβ1-induced stress fiber formation by inhibiting myosin light chain phosphorylation.J Ethnopharmacol. 2014 Mar 14;152(2):364-71. doi: 10.1016/j.jep.2014.01.024. Epub 2014 Jan 30. J Ethnopharmacol. 2014. PMID: 24486209
-
Schisandrin B suppresses TGFβ1 signaling by inhibiting Smad2/3 and MAPK pathways.Biochem Pharmacol. 2012 Feb 1;83(3):378-84. doi: 10.1016/j.bcp.2011.11.002. Epub 2011 Nov 12. Biochem Pharmacol. 2012. PMID: 22100726
-
Transcriptome Analysis of the Anti-TGFβ Effect of Schisandra chinensis Fruit Extract and Schisandrin B in A7r5 Vascular Smooth Muscle Cells.Life (Basel). 2021 Feb 20;11(2):163. doi: 10.3390/life11020163. Life (Basel). 2021. PMID: 33672474 Free PMC article.
-
Effects of Schisandra chinensis extract on the contractility of corpus cavernosal smooth muscle (CSM) and Ca2+ homeostasis in CSM cells.BJU Int. 2012 May;109(9):1404-13. doi: 10.1111/j.1464-410X.2011.10567.x. Epub 2011 Sep 27. BJU Int. 2012. PMID: 21951618
-
The protective effects of Schisandra chinensis fruit extract and its lignans against cardiovascular disease: a review of the molecular mechanisms.Fitoterapia. 2014 Sep;97:224-33. doi: 10.1016/j.fitote.2014.06.014. Epub 2014 Jun 27. Fitoterapia. 2014. PMID: 24976588 Review.
Cited by
-
The traditional Chinese medicines treat chronic heart failure and their main bioactive constituents and mechanisms.Acta Pharm Sin B. 2023 May;13(5):1919-1955. doi: 10.1016/j.apsb.2023.02.005. Epub 2023 Feb 11. Acta Pharm Sin B. 2023. PMID: 37250151 Free PMC article. Review.
-
Immunomodulatory effects of fermented Platycodon grandiflorum extract through NF-κB signaling in RAW 264.7 cells.Nutr Res Pract. 2020 Oct;14(5):453-462. doi: 10.4162/nrp.2020.14.5.453. Epub 2020 Jun 8. Nutr Res Pract. 2020. PMID: 33029286 Free PMC article.
-
Nuciferine attenuates lipopolysaccharide-stimulated inflammatory responses by inhibiting p38 MAPK/ATF2 signaling pathways.Inflammopharmacology. 2022 Dec;30(6):2373-2383. doi: 10.1007/s10787-022-01075-y. Epub 2022 Oct 11. Inflammopharmacology. 2022. PMID: 36219321
-
Polydatin prevents bleomycin-induced pulmonary fibrosis by inhibiting the TGF-β/Smad/ERK signaling pathway.Exp Ther Med. 2020 Nov;20(5):62. doi: 10.3892/etm.2020.9190. Epub 2020 Sep 4. Exp Ther Med. 2020. PMID: 32952652 Free PMC article.
-
HPLC-MS/MS-Mediated Analysis of the Pharmacokinetics, Bioavailability, and Tissue Distribution of Schisandrol B in Rats.Int J Anal Chem. 2021 Feb 24;2021:8862291. doi: 10.1155/2021/8862291. eCollection 2021. Int J Anal Chem. 2021. PMID: 33679983 Free PMC article.
References
-
- Leask A, Abraham DJ. TGF-β signaling and the fibrotic response. FASEB J. 2004;18:816–827. - PubMed
-
- Santibanez JF, Quintanilla M, Bernabeu C. TGF-β/TGF-β receptor system and its role in physiological and pathological conditions. Clin Sci (Lond) 2011;121:233–251. - PubMed
-
- Meng XM, Nikolic-Paterson DJ, Lan HY. TGF-β: the master regulator of fibrosis. Nat Rev Nephrol. 2016;12:325–338. - PubMed
-
- McCaffrey TA. TGF-β signaling in atherosclerosis and restenosis. Front Biosci (Schol Ed) 2009;1:236–245. - PubMed
-
- Ruiz-Ortega M, Rodriguez-Vita J, Sanchez-Lopez E, Carvajal G, Egido J. TGF-β signaling in vascular fibrosis. Cardiovasc Res. 2007;74:196–206. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

