Exceptional potency and structural basis of a T1249-derived lipopeptide fusion inhibitor against HIV-1, HIV-2, and simian immunodeficiency virus
- PMID: 29425101
- PMCID: PMC5892594
- DOI: 10.1074/jbc.RA118.001729
Exceptional potency and structural basis of a T1249-derived lipopeptide fusion inhibitor against HIV-1, HIV-2, and simian immunodeficiency virus
Abstract
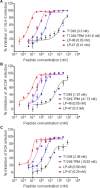
Enfuvirtide (T20) is the only viral fusion inhibitor approved for clinical use, but it has relatively weak anti-HIV activity and easily induces drug resistance. In succession to T20, T1249 has been designed as a 39-mer peptide composed of amino acid sequences derived from HIV-1, HIV-2, and simian immunodeficiency virus (SIV); however, its development has been suspended due to formulation difficulties. We recently developed a T20-based lipopeptide (LP-40) showing greatly improved pharmaceutical properties. Here, we generated a T1249-based lipopeptide, termed LP-46, by replacing its C-terminal tryptophan-rich sequence with fatty acid. As compared with T20, T1249, and LP-40, the truncated LP-46 (31-mer) had dramatically increased activities in inhibiting a large panel of HIV-1 subtypes, with IC50 values approaching low picomolar concentrations. Also, LP-46 was an exceptionally potent inhibitor against HIV-2, SIV, and T20-resistant variants, and it displayed obvious synergistic effects with LP-40. Furthermore, we showed that LP-46 had increased helical stability and binding affinity with the target site. The crystal structure of LP-46 in complex with a target surrogate revealed its critical binding motifs underlying the mechanism of action. Interestingly, it was found that the introduced pocket-binding domain in LP-46 did not interact with the gp41 pocket as expected; instead, it adopted a mode similar to that of LP-40. Therefore, our studies have provided an exceptionally potent and broad fusion inhibitor for developing new anti-HIV drugs, which can also serve as a tool to exploit the mechanisms of viral fusion and inhibition.
Keywords: HIV-1; HIV-2; T1249; T20; antiviral agent; crystal structure; fusion inhibitor; human immunodeficiency virus (HIV); inhibitor; lipopeptide; membrane fusion; peptides; structure-function.
© 2018 Zhu et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures



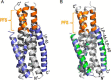

References
-
- Yahi N., Fantini J., Baghdiguian S., Mabrouk K., Tamalet C., Rochat H., Van Rietschoten J., and Sabatier J. M. (1995) SPC3, a synthetic peptide derived from the V3 domain of human immunodeficiency virus type 1 (HIV-1) gp120, inhibits HIV-1 entry into CD4+ and CD4− cells by two distinct mechanisms. Proc. Natl. Acad. Sci. U.S.A. 92, 4867–4871 10.1073/pnas.92.11.4867 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

