Injury, repair, inflammation and metaplasia in the stomach
- PMID: 29427515
- PMCID: PMC6117566
- DOI: 10.1113/JP275512
Injury, repair, inflammation and metaplasia in the stomach
Abstract
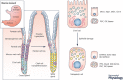
The development of intestinal-type gastric cancer is preceded by the emergence of metaplastic cell lineages in the gastric mucosa. In particular, intestinal metaplasia and spasmolytic polypeptide-expressing metaplasia (SPEM) have been associated with the pathological progression to intestinal-type gastric cancer. The development of SPEM represents a physiological response to damage that recruits reparative cells to sites of mucosal injury. Metaplastic cell lineages are characterized by mucus secretion, adding a protective barrier to the epithelium. Increasing evidence indicates that the influence of alarmins and cytokines is required to initiate the process of metaplasia development. In particular, IL-33 derived from epithelial cells stimulates IL-13 production by specialized innate immune cells to induce chief cell transdifferentiation into SPEM following the loss of parietal cells from the corpus of the stomach. While SPEM represents a physiological healing response to acute injury, persistent injury and chronic inflammation can perpetuate a recurring pattern of reprogramming and metaplasia that is a risk factor for gastric cancer development. The transdifferentiation of zymogen secreting cells into mucous cell metaplasia may represent both a general repair mechanism in response to mucosal injury in many epithelia as well as a common pre-neoplastic pathway associated with chronic injury and inflammation.
Keywords: IL-13; IL-33; SPEM; ST2; macrophage polarization; spasmolytic polypeptide-expressing metaplasia; transdifferentiation.
© 2018 The Authors. The Journal of Physiology © 2018 The Physiological Society.
Figures

References
-
- Bernink JH, Germar K & Spits H (2014). The role of ILC2 in pathology of type 2 inflammatory diseases. Curr Opin Immunol 31, 115–120. - PubMed
-
- Byers DE, Alexander‐Brett J, Patel AC, Agapov E, Dang‐Vu G, Jin X, Wu K, You Y, Alevy Y, Girard JP, Stappenbeck TS, Patterson GA, Pierce RA, Brody SL & Holtzman MJ (2013). Long‐term IL‐33‐producing epithelial progenitor cells in chronic obstructive lung disease. J Clin Invest 123, 3967–3982. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

